QUESTION IMAGE
Question
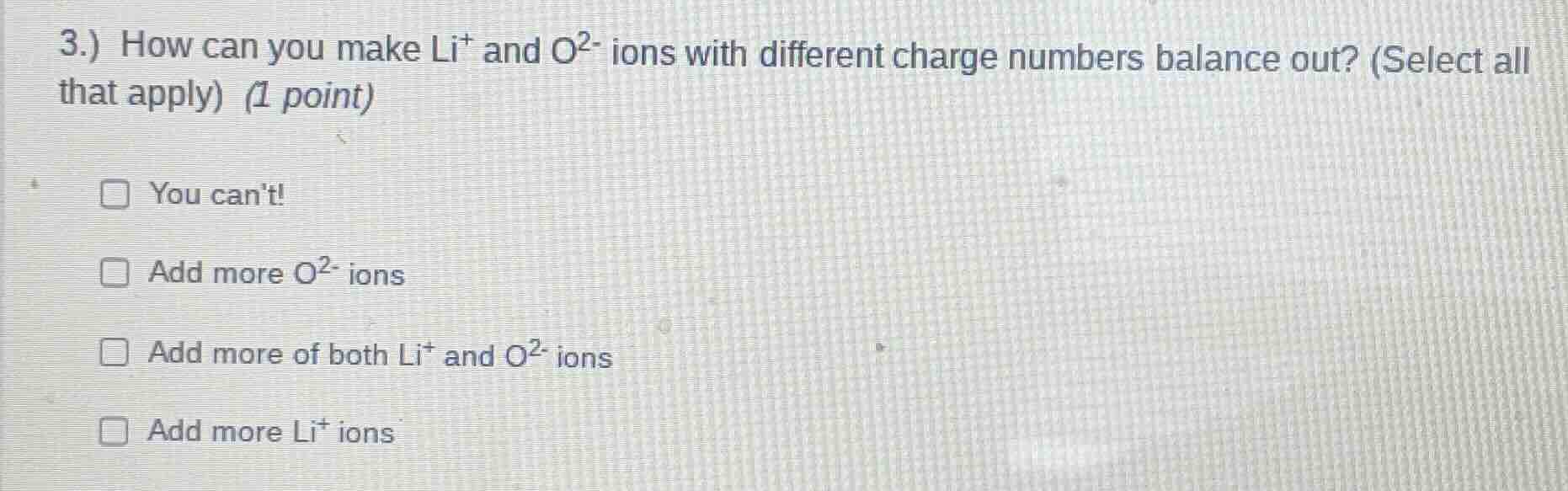
3.) how can you make li⁺ and o²⁻ ions with different charge numbers balance out? (select all that apply) (1 point)
□ you cant!
□ add more o²⁻ ions
□ add more of both li⁺ and o²⁻ ions
□ add more li⁺ ions
To balance the charges of $\text{Li}^+$ (charge +1) and $\text{O}^{2-}$ (charge -2), we need the total positive charge to equal the total negative charge. Let the number of $\text{Li}^+$ ions be $x$ and $\text{O}^{2-}$ ions be $y$. The charge balance equation is $+1\times x + (-2)\times y = 0$, which simplifies to $x = 2y$. This means we need twice as many $\text{Li}^+$ ions as $\text{O}^{2-}$ ions. So, we can balance them by adding more $\text{Li}^+$ ions (to get the ratio right) or adding more of both in the correct ratio. "You can't!" is wrong because we can balance them. "Add more $\text{O}^{2-}$ ions" would make the negative charge larger, unbalancing it more. "Add more $\text{Li}^+$ ions" helps reach the $x = 2y$ ratio. "Add more of both $\text{Li}^+$ and $\text{O}^{2-}$ ions" (in the ratio 2:1) also works.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Add more $\text{Li}^+$ ions, C. Add more of both $\text{Li}^+$ and $\text{O}^{2-}$ ions (Note: Assuming the options are labeled as follows: A. You can't!, B. Add more $\text{O}^{2-}$ ions, C. Add more of both $\text{Li}^+$ and $\text{O}^{2-}$ ions, D. Add more $\text{Li}^+$ ions)