QUESTION IMAGE
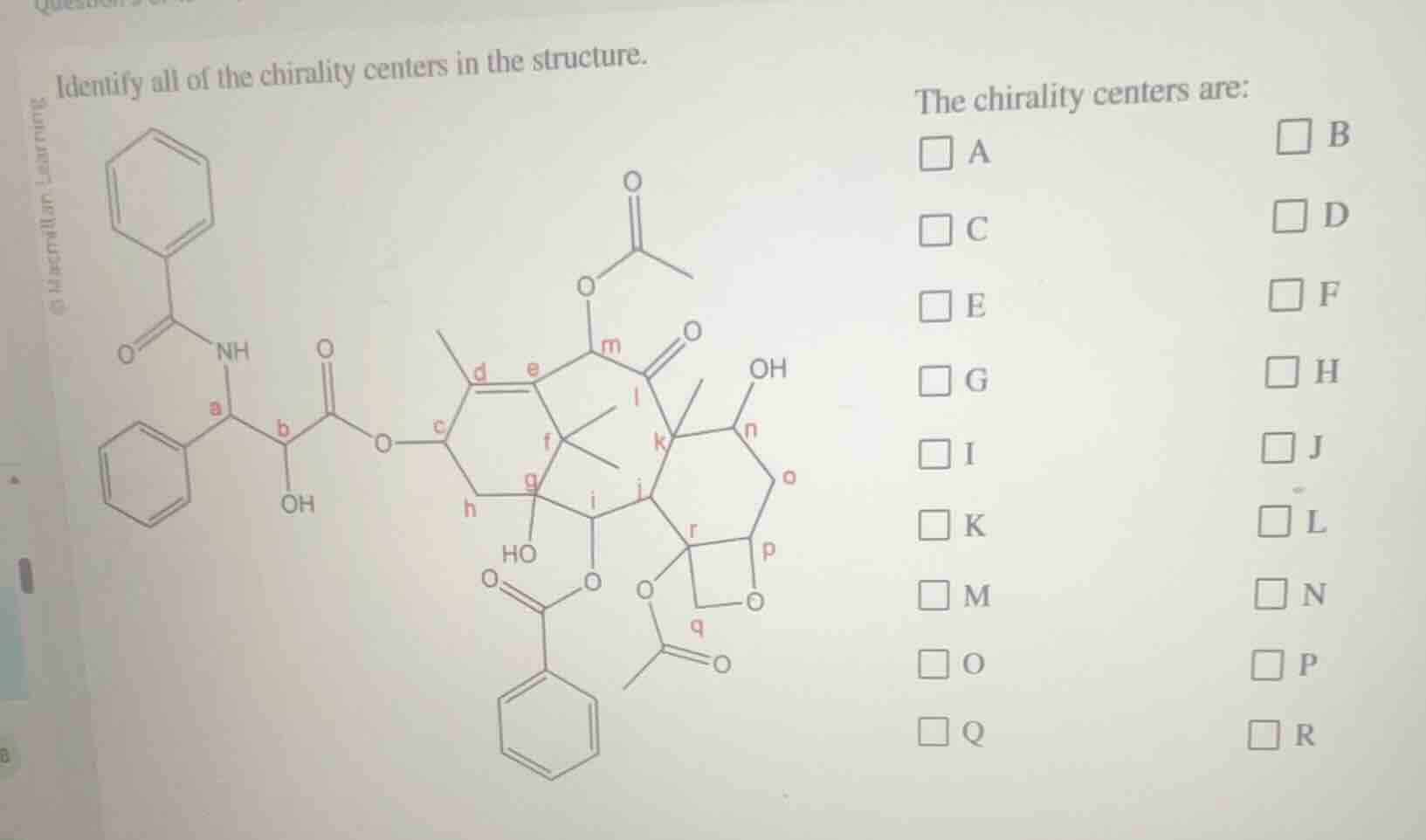
Question
identify all of the chirality centers in the structure.
the chirality centers are:
□ a □ b
□ c □ d
□ e □ f
□ g □ h
□ i □ j
□ k □ l
□ m □ n
□ o □ p
□ q □ r
Step1: Define chirality center
A chirality center is a tetrahedral atom (usually C) bonded to 4 distinct groups; sp² C (double-bonded) is excluded.
Step2: Evaluate each labeled atom
- A: C bonded to C₆H₅, H, NHCOC₆H₅, and group B → 4 distinct groups: chiral.
- B: C bonded to OH, H, group A, and COO-group C → 4 distinct groups: chiral.
- C: C bonded to O-ester, H, group H, and group D → 4 distinct groups: chiral.
- D: sp² C (double bond) → not chiral.
- E: sp² C (double bond) → not chiral.
- F: C bonded to 3 identical CH₃ groups → not chiral.
- G: C bonded to OH, group H, group I, and two CH₃ (on F) → 4 distinct groups: chiral.
- H: C bonded to group C, group G, H, and ring C → 4 distinct groups: chiral.
- I: C bonded to OCOC₆H₅, group G, group J, and H → 4 distinct groups: chiral.
- J: C bonded to group I, group K, and ring C → 4 distinct groups: chiral.
- K: C bonded to 2 CH₃ groups, group J, and group N → not chiral.
- L: sp² C (carbonyl) → not chiral.
- M: C bonded to OAc, group E, group L, and H → 4 distinct groups: chiral.
- N: C bonded to OH, group K, group O, and H → 4 distinct groups: chiral.
- O: C bonded to group N, group P, H, and O → 4 distinct groups: chiral.
- P: C bonded to group O, group R, and 2 O atoms → 4 distinct groups: chiral.
- Q: sp² C (carbonyl) → not chiral.
- R: C bonded to OAc, group J, group P, and ring C → 4 distinct groups: chiral.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. A, B. B, C. C, G. G, H. H, I. I, J. J, M. M, N. N, O. O, P. P, R. R