QUESTION IMAGE
Question
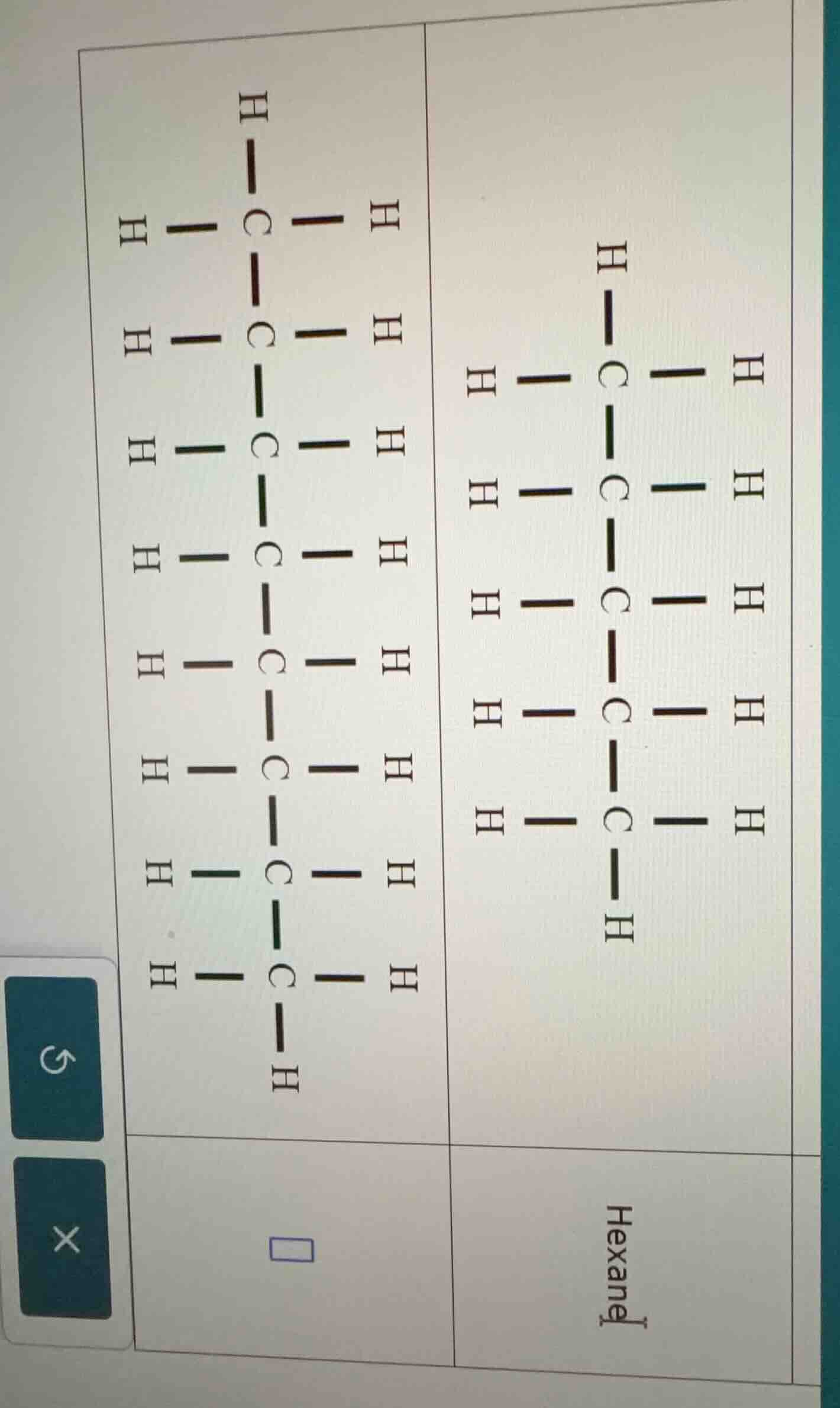
(the image shows two structural formulas of hydrocarbons, one is hexane on the right, and the left one is a hydrocarbon structure with more carbon atoms. there is a blank box below the left structure.)
Step1: Count Carbon atoms in left structure
Looking at the left Lewis structure, we count the number of C atoms. By examining the bonds, we see there are 9 C atoms (each C is connected as per the structure: let's list them - the chain has C atoms: first C, then connected to next C, and so on, totaling 9 C atoms).
Step2: Determine the alkane name
Alkanes have the formula $C_nH_{2n + 2}$. For $n = 9$, the alkane is nonane. Let's verify the H atoms: for $n = 9$, $H = 2(9)+2 = 20$. Looking at the structure, each C (except the ends) has 2 H, and the ends have 3 H? Wait, no, in the left structure, let's count H: each internal C (between two C - C bonds) has 2 H, and the terminal C (first and last) have 3 H? Wait, no, the left structure: let's count C atoms. Let's see the bonds: the first C is bonded to 3 H and 1 C, the next 7 C atoms (between first and last) are bonded to 2 H and 2 C (except maybe? Wait, no, the left structure: let's count the number of C - C bonds. Let's see the vertical chain: how many C? Let's count the C atoms: from top to bottom, the C atoms are: 1 (top), then 2, 3, 4, 5, 6, 7, 8, 9 (bottom). So 9 C atoms. So the alkane is nonane. The right structure is hexane (6 C atoms). So the left structure is nonane.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Nonane (the left structure is nonane, with 9 carbon atoms, formula $C_9H_{20}$)