QUESTION IMAGE
Question
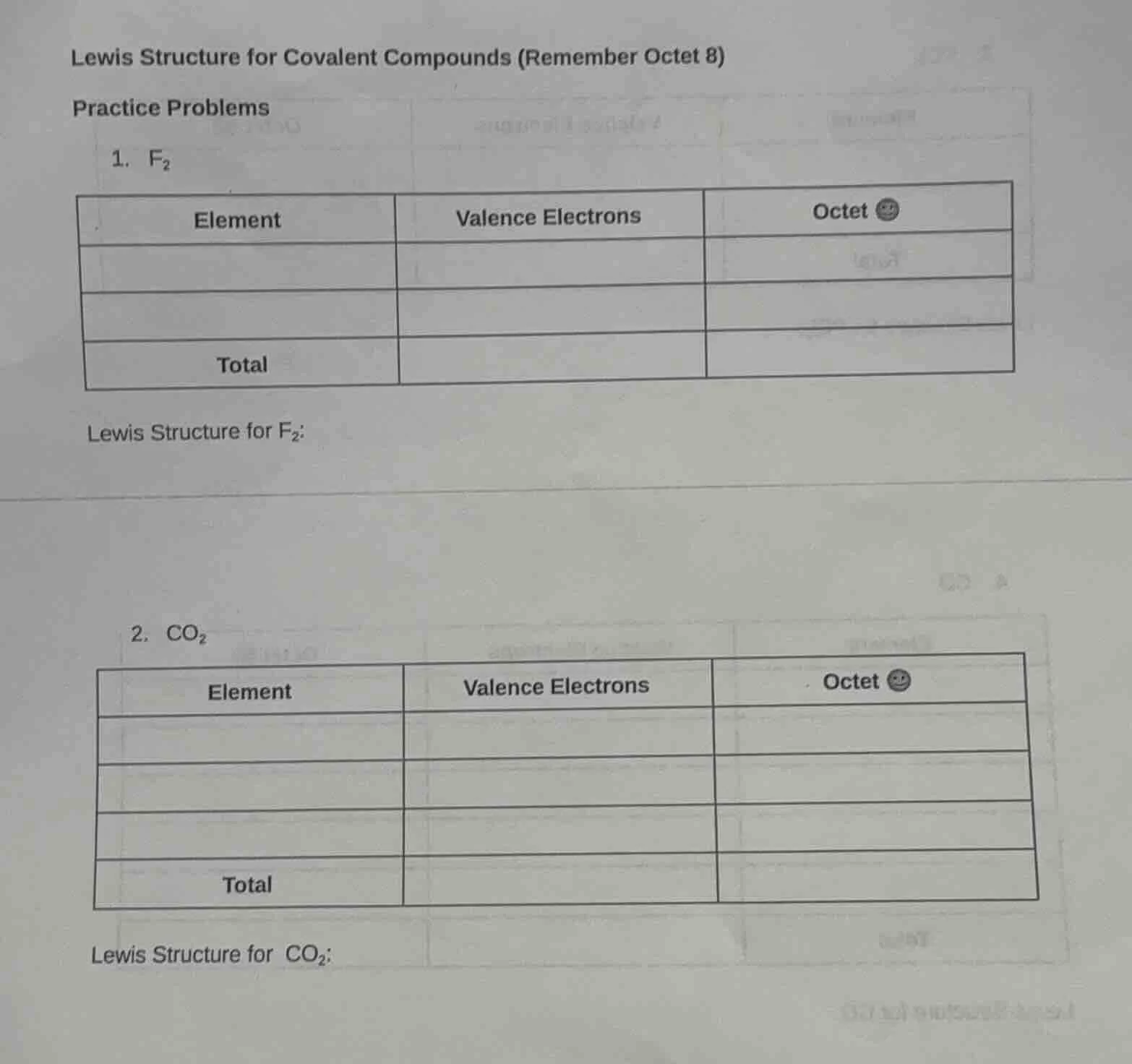
lewis structure for covalent compounds (remember octet 8)
practice problems
- $\ce{f_{2}}$
| element | valence electrons | octet 😀 |
|---|---|---|
| total |
lewis structure for $\ce{f_{2}}$:
- $\ce{co_{2}}$
| element | valence electrons | octet 😀 |
|---|---|---|
| total |
lewis structure for $\ce{co_{2}}$:
Problem 1: \( \boldsymbol{F_2} \)
Step 1: Determine Valence Electrons for Fluorine
Fluorine (F) is in group 17, so it has 7 valence electrons. Since there are two F atoms in \( F_2 \):
- First F atom: Valence electrons = 7, Octet needed = 8 (to satisfy octet rule)
- Second F atom: Valence electrons = 7, Octet needed = 8
Step 2: Calculate Total Valence Electrons and Octet
- Total valence electrons: \( 7 + 7 = 14 \)
- Total octet needed: \( 8 + 8 = 16 \) (but we use the bonding to share electrons, so the shared pair counts for both)
Filling the Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| F | 7 | 8 |
| Total | \( 7 + 7 = 14 \) | \( 8 + 8 = 16 \) |
Lewis Structure for \( \boldsymbol{F_2} \)
- Each F atom contributes 7 valence electrons. They share a pair of electrons (a single bond), so the Lewis structure is: \( \text{F} - \text{F} \), with 3 lone pairs on each F (since \( 7 - 1 = 6 \) non - bonding electrons, which form 3 lone pairs).
Problem 2: \( \boldsymbol{CO_2} \)
Step 1: Determine Valence Electrons for Each Element
- Carbon (C) is in group 14, so it has 4 valence electrons. Octet needed = 8.
- Oxygen (O) is in group 16, so each O has 6 valence electrons. There are 2 O atoms, and each O needs 8 electrons for octet.
Step 2: Calculate Total Valence Electrons and Octet
- Total valence electrons: \( 4+(6\times2)=4 + 12 = 16 \)
- Total octet needed: \( 8+(8\times2)=8 + 16 = 24 \)
Filling the Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| O | 6 | 8 |
| O | 6 | 8 |
| Total | \( 4+6 + 6=16 \) | \( 8 + 8+8 = 24 \) |
Lewis Structure for \( \boldsymbol{CO_2} \)
- Carbon is the central atom. It forms double bonds with each O atom. The valence electrons: C has 4, each O has 6. The Lewis structure is \( \text{O}=\text{C}=\text{O} \). Each O has 2 lone pairs (since \( 6 - 2 = 4 \) non - bonding electrons, forming 2 lone pairs) and C has no lone pairs (as it uses all 4 electrons in double bonds to satisfy the octet).
Final Answers (Table Fills and Lewis Structures)
For \( \boldsymbol{F_2} \) Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| F | 7 | 8 |
| Total | 14 | 16 |
Lewis Structure: \( \text{F} - \text{F} \) (with 3 lone pairs on each F)
For \( \boldsymbol{CO_2} \) Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| O | 6 | 8 |
| O | 6 | 8 |
| Total | 16 | 24 |
Lewis Structure: \( \text{O}=\text{C}=\text{O} \) (with 2 lone pairs on each O)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Problem 1: \( \boldsymbol{F_2} \)
Step 1: Determine Valence Electrons for Fluorine
Fluorine (F) is in group 17, so it has 7 valence electrons. Since there are two F atoms in \( F_2 \):
- First F atom: Valence electrons = 7, Octet needed = 8 (to satisfy octet rule)
- Second F atom: Valence electrons = 7, Octet needed = 8
Step 2: Calculate Total Valence Electrons and Octet
- Total valence electrons: \( 7 + 7 = 14 \)
- Total octet needed: \( 8 + 8 = 16 \) (but we use the bonding to share electrons, so the shared pair counts for both)
Filling the Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| F | 7 | 8 |
| Total | \( 7 + 7 = 14 \) | \( 8 + 8 = 16 \) |
Lewis Structure for \( \boldsymbol{F_2} \)
- Each F atom contributes 7 valence electrons. They share a pair of electrons (a single bond), so the Lewis structure is: \( \text{F} - \text{F} \), with 3 lone pairs on each F (since \( 7 - 1 = 6 \) non - bonding electrons, which form 3 lone pairs).
Problem 2: \( \boldsymbol{CO_2} \)
Step 1: Determine Valence Electrons for Each Element
- Carbon (C) is in group 14, so it has 4 valence electrons. Octet needed = 8.
- Oxygen (O) is in group 16, so each O has 6 valence electrons. There are 2 O atoms, and each O needs 8 electrons for octet.
Step 2: Calculate Total Valence Electrons and Octet
- Total valence electrons: \( 4+(6\times2)=4 + 12 = 16 \)
- Total octet needed: \( 8+(8\times2)=8 + 16 = 24 \)
Filling the Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| O | 6 | 8 |
| O | 6 | 8 |
| Total | \( 4+6 + 6=16 \) | \( 8 + 8+8 = 24 \) |
Lewis Structure for \( \boldsymbol{CO_2} \)
- Carbon is the central atom. It forms double bonds with each O atom. The valence electrons: C has 4, each O has 6. The Lewis structure is \( \text{O}=\text{C}=\text{O} \). Each O has 2 lone pairs (since \( 6 - 2 = 4 \) non - bonding electrons, forming 2 lone pairs) and C has no lone pairs (as it uses all 4 electrons in double bonds to satisfy the octet).
Final Answers (Table Fills and Lewis Structures)
For \( \boldsymbol{F_2} \) Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| F | 7 | 8 |
| Total | 14 | 16 |
Lewis Structure: \( \text{F} - \text{F} \) (with 3 lone pairs on each F)
For \( \boldsymbol{CO_2} \) Table:
| Element | Valence Electrons | Octet |
|---|---|---|
| O | 6 | 8 |
| O | 6 | 8 |
| Total | 16 | 24 |
Lewis Structure: \( \text{O}=\text{C}=\text{O} \) (with 2 lone pairs on each O)