QUESTION IMAGE
Question
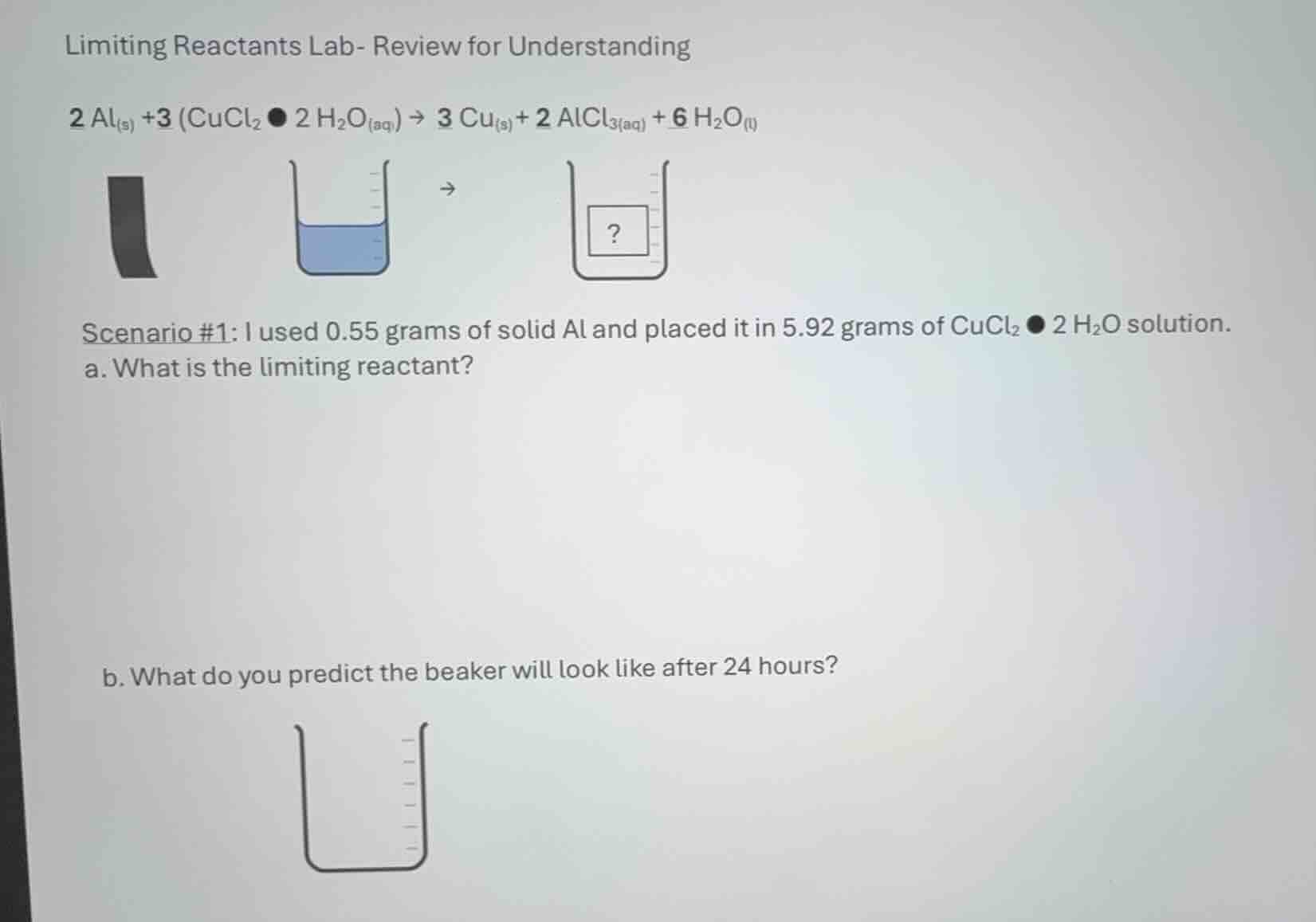
limiting reactants lab- review for understanding
$ce{2 al_{(s)} +3 (cucl_{2}•2 h_{2}o_{(aq)}) -> 3 cu_{(s)} + 2 alcl_{3(aq)} + 6 h_{2}o_{(l)}}$
scenario #1: i used 0.55 grams of solid al and placed it in 5.92 grams of $ce{cucl_{2}•2 h_{2}o}$ solution.
a. what is the limiting reactant?
b. what do you predict the beaker will look like after 24 hours?
Step1: Calculate molar mass of Al
Molar mass of $\text{Al} = 26.98\ \text{g/mol}$
Step2: Calculate moles of Al
$\text{Moles of Al} = \frac{0.55\ \text{g}}{26.98\ \text{g/mol}} \approx 0.0204\ \text{mol}$
Step3: Calculate molar mass of $\text{CuCl}_2\cdot2\text{H}_2\text{O}$
$\text{Molar mass} = 63.55 + 2\times35.45 + 2\times(2\times1.01 + 16.00) = 170.48\ \text{g/mol}$
Step4: Calculate moles of $\text{CuCl}_2\cdot2\text{H}_2\text{O}$
$\text{Moles of }\text{CuCl}_2\cdot2\text{H}_2\text{O} = \frac{5.92\ \text{g}}{170.48\ \text{g/mol}} \approx 0.0347\ \text{mol}$
Step5: Find mole ratio from reaction
Reaction ratio: $\frac{\text{mol Al}}{\text{mol }\text{CuCl}_2\cdot2\text{H}_2\text{O}} = \frac{2}{3} \approx 0.667$
Step6: Calculate actual mole ratio
Actual ratio: $\frac{0.0204}{0.0347} \approx 0.588$
Step7: Compare ratios to find limiting reactant
Actual ratio < Reaction ratio, so Al is in excess, $\text{CuCl}_2\cdot2\text{H}_2\text{O}$ is limiting.
(Part b):
The limiting reactant $\text{CuCl}_2\cdot2\text{H}_2\text{O}$ will be fully consumed. The blue color of the $\text{CuCl}_2$ solution will disappear, replaced by a colorless solution of $\text{AlCl}_3$. Solid reddish-brown copper metal will precipitate at the bottom of the beaker, and some unreacted solid aluminum will remain (since Al was in excess).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Part a):
The limiting reactant is $\text{CuCl}_2\cdot2\text{H}_2\text{O}$ (copper(II) chloride dihydrate).