QUESTION IMAGE
Question
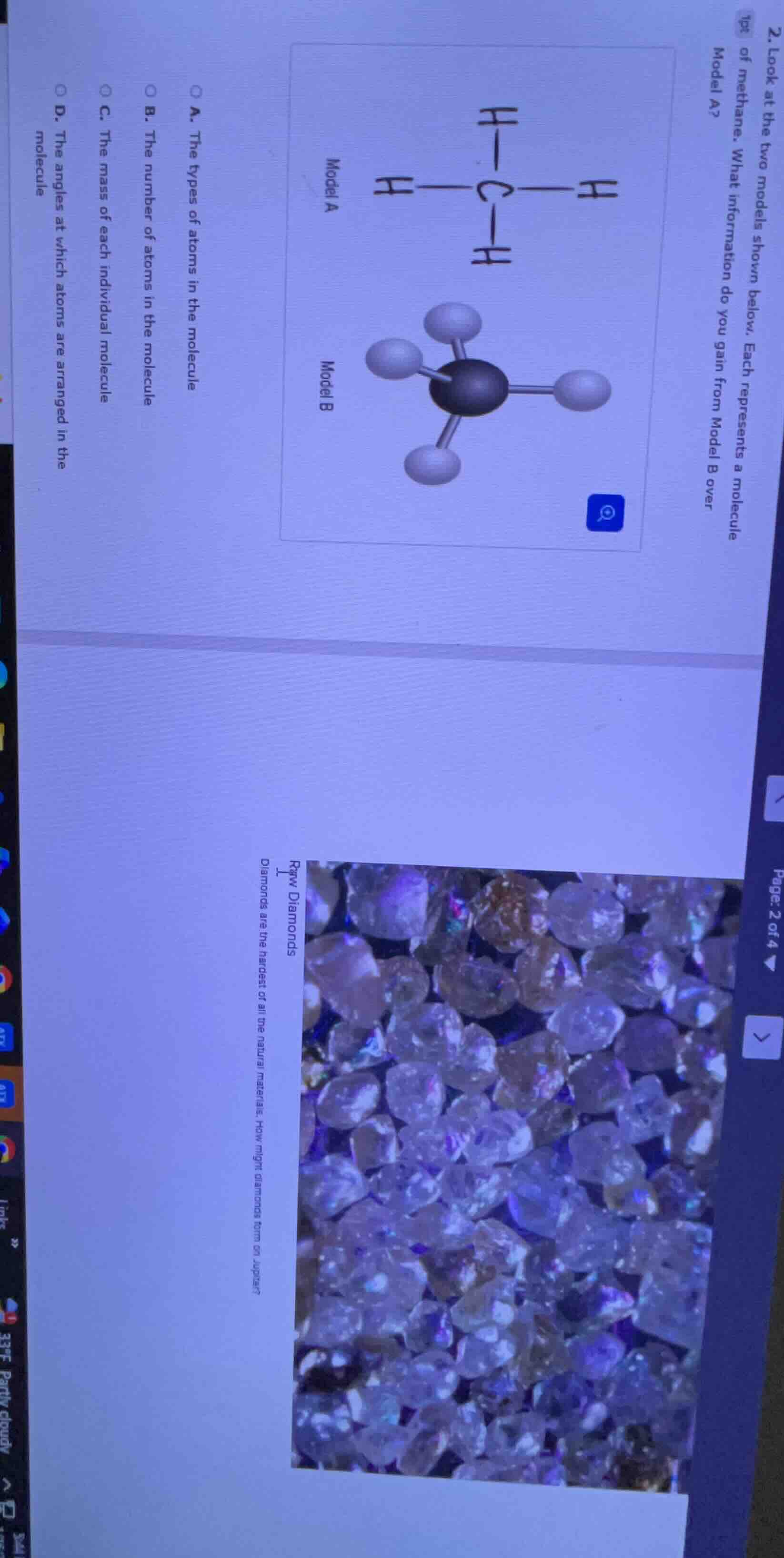
- look at the two models shown below. each represents a molecule of methane. what information do you gain from model b over model a?
model a
model b
a. the types of atoms in the molecule
b. the number of atoms in the molecule
c. the mass of each individual molecule
d. the angles at which atoms are arranged in the molecule
raw diamonds
diamonds are the hardest of all the natural materials. how might diamonds form so pure?
Brief Explanations
- Option A: Both Model A (Lewis structure) and Model B (ball - and - stick model) can show the types of atoms (C and H in methane), so this is not unique to Model B.
- Option B: Both models can show the number of atoms (1 C and 4 H in methane), so this is not unique to Model B.
- Option C: Neither model directly shows the mass of each molecule, as mass is related to atomic masses and requires calculation, not direct visualization from these models.
- Option D: Model A (Lewis structure) is a 2 - D representation and does not show the bond angles or the 3 - D arrangement angles of atoms. Model B (ball - and - stick model) is a 3 - D representation that can show the angles at which atoms are arranged in the molecule (the tetrahedral angle in methane's case).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. The angles at which atoms are arranged in the molecule