QUESTION IMAGE
Question
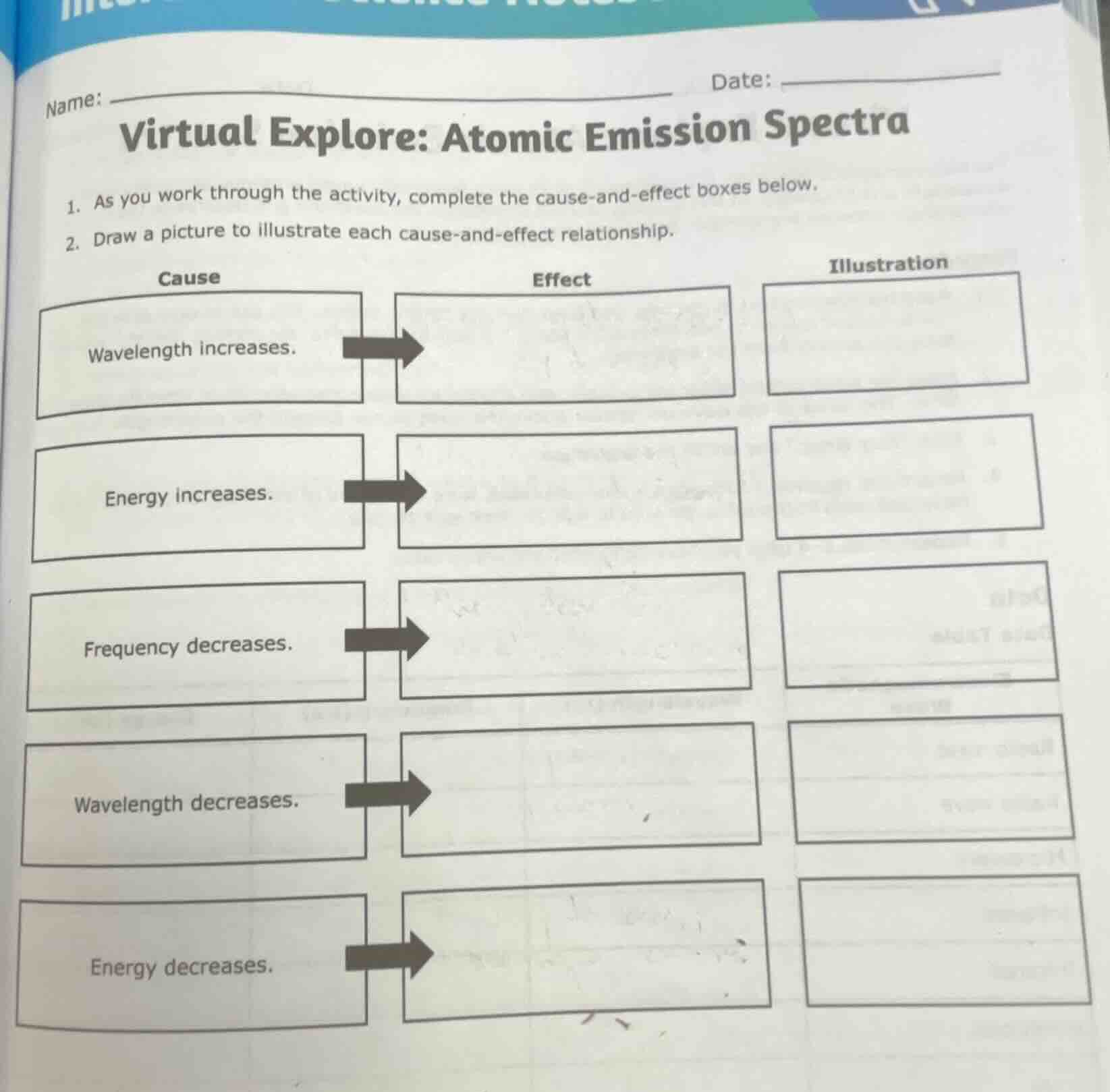
name: date: virtual explore: atomic emission spectra 1. as you work through the activity, complete the cause-and-effect boxes below. 2. draw a picture to illustrate each cause-and-effect relationship. cause effect illustration wavelength increases. energy increases. frequency decreases. wavelength decreases. energy decreases.
To solve the cause - effect relationships related to atomic emission spectra, we use the fundamental relationships between wavelength ($\lambda$), frequency ($f$), and energy ($E$) of electromagnetic radiation. The key formulas are $c=\lambda f$ (where $c$ is the speed of light in a vacuum, a constant) and $E = hf$ (where $h$ is Planck's constant, also a constant).
For "Wavelength increases"
Step 1: Recall the relationship $c=\lambda f$
Since $c$ is constant, if $\lambda$ (wavelength) increases, from $f=\frac{c}{\lambda}$, we can see that frequency ($f$) will decrease. Also, from $E = hf$, as frequency decreases, energy ($E$) will decrease. But for the direct effect of wavelength increase, using $c=\lambda f$ (with $c$ constant), when $\lambda$ increases, $f=\frac{c}{\lambda}$ so $f$ decreases. And from $E = hf$, $E$ decreases. But the most direct effect from $\lambda$ increase (using $c=\lambda f$) is that frequency decreases and energy (from $E = hf$) also decreases. But if we consider the effect on energy via wavelength, we can also use $E=\frac{hc}{\lambda}$, so as $\lambda$ increases, $E$ decreases. So the effect of "Wavelength increases" is "Frequency decreases and Energy decreases" (or more precisely, from the formula $E=\frac{hc}{\lambda}$, as $\lambda$ increases, $E$ decreases; and from $c = \lambda f$, as $\lambda$ increases, $f$ decreases).
For "Energy increases"
Step 1: Recall the formula $E = hf$
Since $h$ is constant, if $E$ (energy) increases, then from $f=\frac{E}{h}$, frequency ($f$) must increase. And from $c=\lambda f$, with $c$ constant, if $f$ increases, then $\lambda=\frac{c}{f}$ so wavelength ($\lambda$) decreases. So the effect of "Energy increases" is "Frequency increases and Wavelength decreases".
For "Frequency decreases"
Step 1: Recall the formula $E = hf$
Since $h$ is constant, if $f$ (frequency) decreases, then from $E = hf$, energy ($E$) decreases. Also, from $c=\lambda f$, with $c$ constant, if $f$ decreases, then $\lambda=\frac{c}{f}$ so wavelength ($\lambda$) increases. So the effect of "Frequency decreases" is "Energy decreases and Wavelength increases".
For "Wavelength decreases"
Step 1: Recall the formula $c=\lambda f$
Since $c$ is constant, if $\lambda$ (wavelength) decreases, then from $f=\frac{c}{\lambda}$, frequency ($f$) increases. And from $E = hf$, as $f$ increases, energy ($E$) increases. So the effect of "Wavelength decreases" is "Frequency increases and Energy increases".
For "Energy decreases"
Step 1: Recall the formula $E = hf$
Since $h$ is constant, if $E$ (energy) decreases, then from $f=\frac{E}{h}$, frequency ($f$) decreases. And from $c=\lambda f$, with $c$ constant, if $f$ decreases, then $\lambda=\frac{c}{f}$ so wavelength ($\lambda$) increases. So the effect of "Energy decreases" is "Frequency decreases and Wavelength increases".
Summary of Effects:
- Cause: Wavelength increases; Effect: Frequency decreases, Energy decreases (from $E=\frac{hc}{\lambda}$ and $c = \lambda f$)
- Cause: Energy increases; Effect: Frequency increases, Wavelength decreases (from $E = hf$ and $c=\lambda f$)
- Cause: Frequency decreases; Effect: Energy decreases, Wavelength increases (from $E = hf$ and $c=\lambda f$)
- Cause: Wavelength decreases; Effect: Frequency increases, Energy increases (from $c=\lambda f$ and $E = hf$)
- Cause: Energy decreases; Effect: Frequency decreases, Wavelength increases (from $E = hf$ and $c=\lambda f$)
If we want to fill in the "Effect" boxes with the most direct relationship (using the primary formulas):
- Wavelength incre…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve the cause - effect relationships related to atomic emission spectra, we use the fundamental relationships between wavelength ($\lambda$), frequency ($f$), and energy ($E$) of electromagnetic radiation. The key formulas are $c=\lambda f$ (where $c$ is the speed of light in a vacuum, a constant) and $E = hf$ (where $h$ is Planck's constant, also a constant).
For "Wavelength increases"
Step 1: Recall the relationship $c=\lambda f$
Since $c$ is constant, if $\lambda$ (wavelength) increases, from $f=\frac{c}{\lambda}$, we can see that frequency ($f$) will decrease. Also, from $E = hf$, as frequency decreases, energy ($E$) will decrease. But for the direct effect of wavelength increase, using $c=\lambda f$ (with $c$ constant), when $\lambda$ increases, $f=\frac{c}{\lambda}$ so $f$ decreases. And from $E = hf$, $E$ decreases. But the most direct effect from $\lambda$ increase (using $c=\lambda f$) is that frequency decreases and energy (from $E = hf$) also decreases. But if we consider the effect on energy via wavelength, we can also use $E=\frac{hc}{\lambda}$, so as $\lambda$ increases, $E$ decreases. So the effect of "Wavelength increases" is "Frequency decreases and Energy decreases" (or more precisely, from the formula $E=\frac{hc}{\lambda}$, as $\lambda$ increases, $E$ decreases; and from $c = \lambda f$, as $\lambda$ increases, $f$ decreases).
For "Energy increases"
Step 1: Recall the formula $E = hf$
Since $h$ is constant, if $E$ (energy) increases, then from $f=\frac{E}{h}$, frequency ($f$) must increase. And from $c=\lambda f$, with $c$ constant, if $f$ increases, then $\lambda=\frac{c}{f}$ so wavelength ($\lambda$) decreases. So the effect of "Energy increases" is "Frequency increases and Wavelength decreases".
For "Frequency decreases"
Step 1: Recall the formula $E = hf$
Since $h$ is constant, if $f$ (frequency) decreases, then from $E = hf$, energy ($E$) decreases. Also, from $c=\lambda f$, with $c$ constant, if $f$ decreases, then $\lambda=\frac{c}{f}$ so wavelength ($\lambda$) increases. So the effect of "Frequency decreases" is "Energy decreases and Wavelength increases".
For "Wavelength decreases"
Step 1: Recall the formula $c=\lambda f$
Since $c$ is constant, if $\lambda$ (wavelength) decreases, then from $f=\frac{c}{\lambda}$, frequency ($f$) increases. And from $E = hf$, as $f$ increases, energy ($E$) increases. So the effect of "Wavelength decreases" is "Frequency increases and Energy increases".
For "Energy decreases"
Step 1: Recall the formula $E = hf$
Since $h$ is constant, if $E$ (energy) decreases, then from $f=\frac{E}{h}$, frequency ($f$) decreases. And from $c=\lambda f$, with $c$ constant, if $f$ decreases, then $\lambda=\frac{c}{f}$ so wavelength ($\lambda$) increases. So the effect of "Energy decreases" is "Frequency decreases and Wavelength increases".
Summary of Effects:
- Cause: Wavelength increases; Effect: Frequency decreases, Energy decreases (from $E=\frac{hc}{\lambda}$ and $c = \lambda f$)
- Cause: Energy increases; Effect: Frequency increases, Wavelength decreases (from $E = hf$ and $c=\lambda f$)
- Cause: Frequency decreases; Effect: Energy decreases, Wavelength increases (from $E = hf$ and $c=\lambda f$)
- Cause: Wavelength decreases; Effect: Frequency increases, Energy increases (from $c=\lambda f$ and $E = hf$)
- Cause: Energy decreases; Effect: Frequency decreases, Wavelength increases (from $E = hf$ and $c=\lambda f$)
If we want to fill in the "Effect" boxes with the most direct relationship (using the primary formulas):
- Wavelength increases: Effect - Frequency decreases, Energy decreases (from $E=\frac{hc}{\lambda}$ and $c=\lambda f$)
- Energy increases: Effect - Frequency increases, Wavelength decreases (from $E = hf$ and $c=\lambda f$)
- Frequency decreases: Effect - Energy decreases, Wavelength increases (from $E = hf$ and $c=\lambda f$)
- Wavelength decreases: Effect - Frequency increases, Energy increases (from $c=\lambda f$ and $E = hf$)
- Energy decreases: Effect - Frequency decreases, Wavelength increases (from $E = hf$ and $c=\lambda f$)
Final Answers for Each Cause - Effect:
- Cause: Wavelength increases; Effect: Frequency decreases, Energy decreases
- Cause: Energy increases; Effect: Frequency increases, Wavelength decreases
- Cause: Frequency decreases; Effect: Energy decreases, Wavelength increases
- Cause: Wavelength decreases; Effect: Frequency increases, Energy increases
- Cause: Energy decreases; Effect: Frequency decreases, Wavelength increases