QUESTION IMAGE
Question
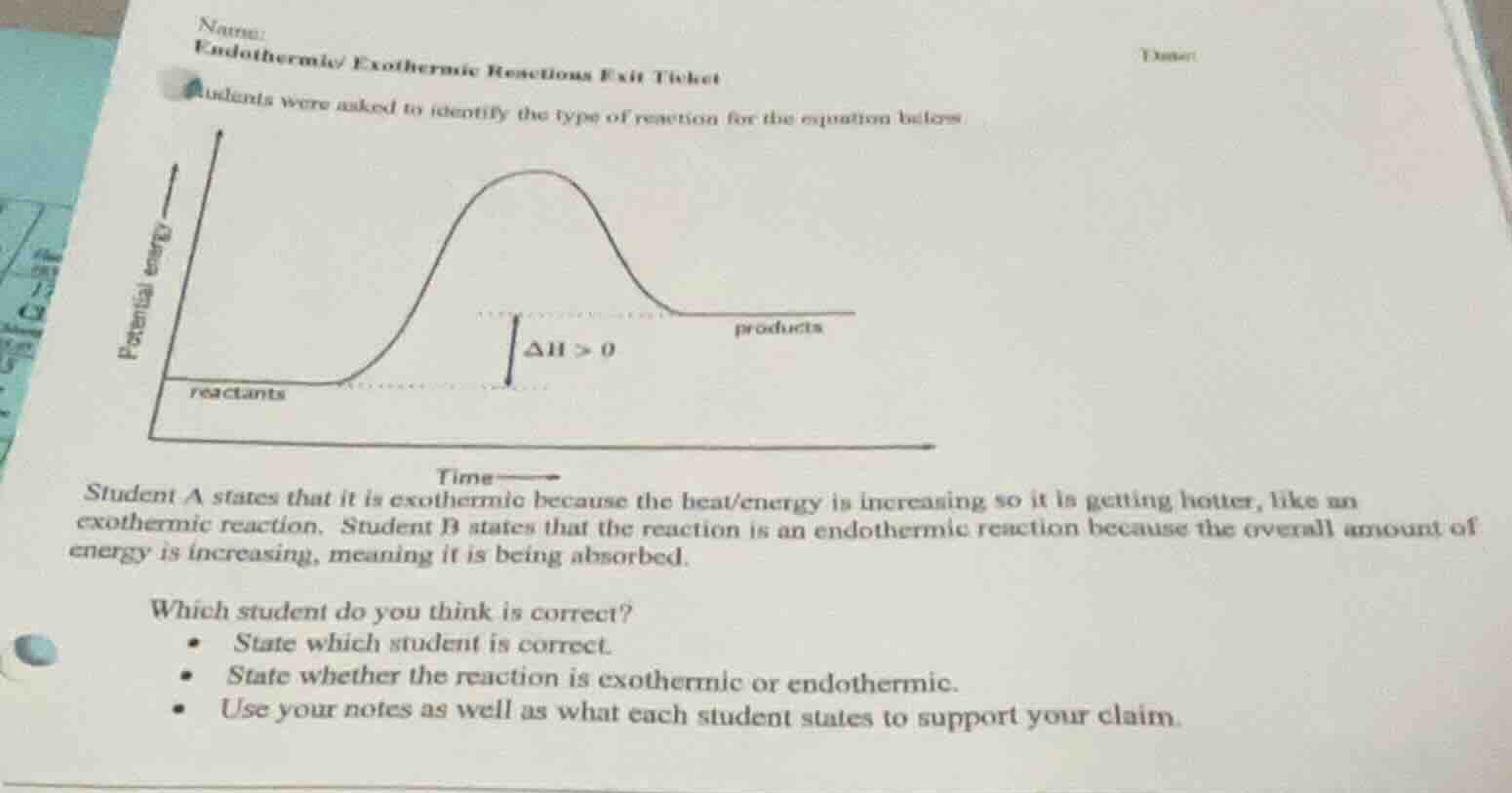
name: endothermic/exothermic reactions exit ticket date: students were asked to identify the type of reaction for the equation below graph of potential energy vs time with reactants, products, δh > 0 student a states that it is exothermic because the heat/energy is increasing so it is getting hotter, like an exothermic reaction. student b states that the reaction is an endothermic reaction because the overall amount of energy is increasing, meaning it is being absorbed. which student do you think is correct? - state which student is correct. - state whether the reaction is exothermic or endothermic. - use your notes as well as what each student states to support your claim.
- For endothermic reactions, the potential energy of products is higher than reactants, so $\Delta H > 0$, meaning the system absorbs energy from the surroundings.
- Student A is wrong: the graph shows products have more energy, so the system takes in heat (it does not release heat/get hotter, which is exothermic).
- Student B is correct: the overall energy of the system increases, which matches the definition of an endothermic reaction (energy is absorbed).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Correct student: Student B
- Reaction type: Endothermic
- Support: The energy graph shows products have higher potential energy than reactants, with $\Delta H > 0$. This means the reaction absorbs energy from the surroundings, which aligns with Student B's reasoning. Student A confused the energy change of the system; an endothermic reaction cools the surroundings (does not make it hotter) as the system takes in energy.