QUESTION IMAGE
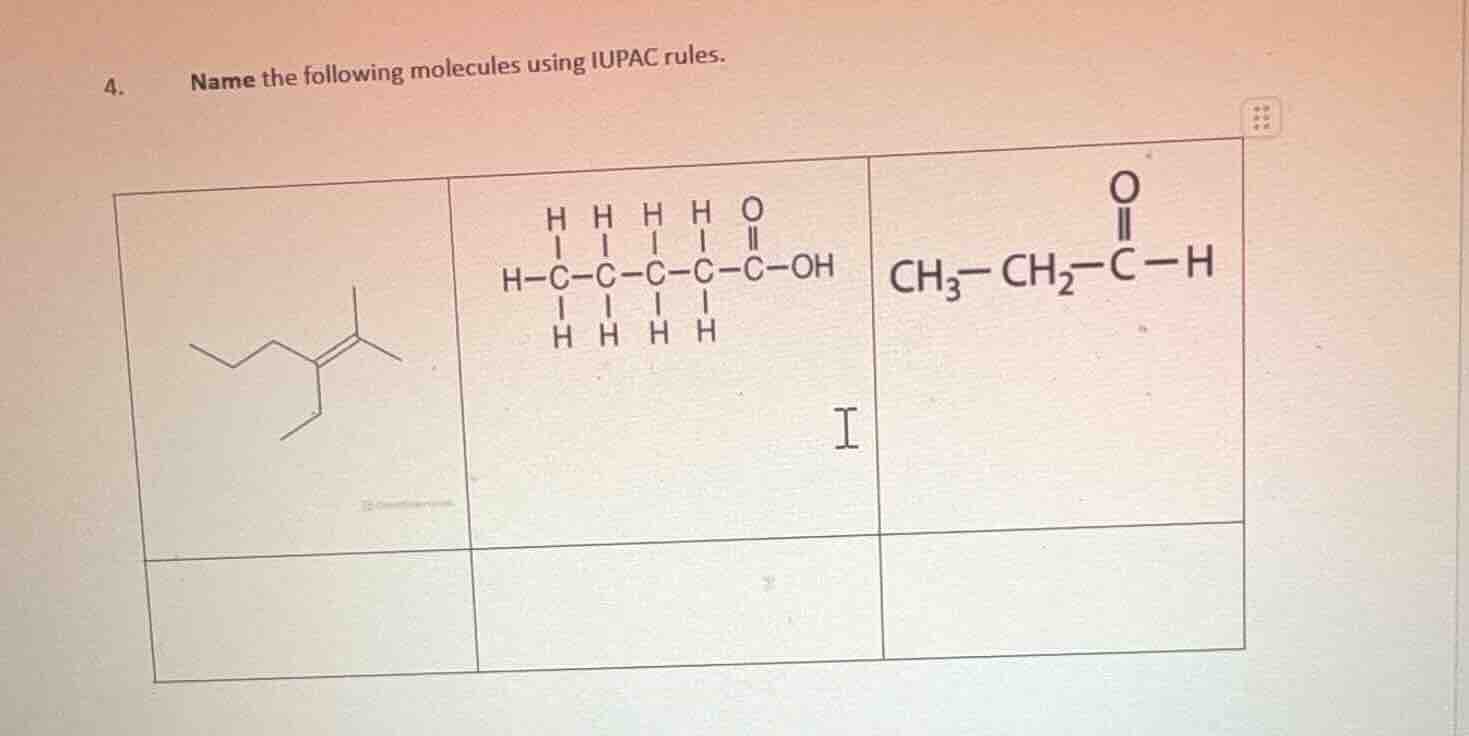
Question
- name the following molecules using iupac rules.
Response
First Molecule (Alkene)
Step1: Find the longest carbon chain with the double bond.
The longest chain has 6 carbons (hexene). The double bond is at carbon 3. Substituents: ethyl at C - 3, methyl at C - 2.
Step2: Number the chain to give the double bond the lowest number, then list substituents alphabetically.
Name: 3 - ethyl - 2 - methyl - 3 - hexene
Second Molecule (Carboxylic Acid)
Step1: Identify the functional group (carboxylic acid, -COOH) and the longest carbon chain.
Longest chain: 5 carbons (pentanoic acid). No substituents.
Step2: Name based on the chain length and functional group.
Name: Pentanoic acid (or 1 - pentanoic acid, but usually pentanoic acid for straight - chain)
Third Molecule (Aldehyde)
Step1: Identify the functional group (aldehyde, -CHO) and the longest carbon chain.
Longest chain: 3 carbons (propanal). No substituents.
Step2: Name based on the chain length and functional group.
Name: Propanal
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- First Molecule: 3 - ethyl - 2 - methyl - 3 - hexene
- Second Molecule: Pentanoic acid
- Third Molecule: Propanal