QUESTION IMAGE
Question
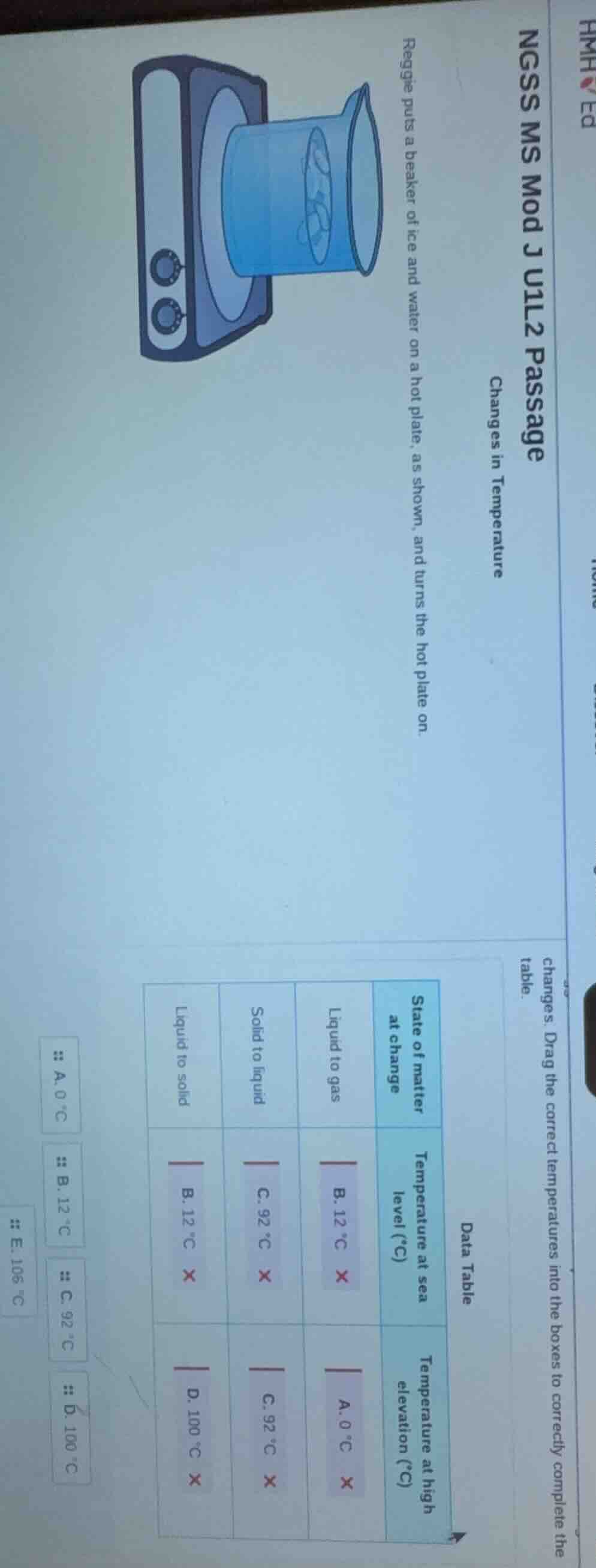
ngss ms mod j u1l2 passage
changes in temperature
reggie puts a beaker of ice and water on a hot plate, as shown, and turns the hot plate on.
changes. drag the correct temperatures into the boxes to correctly complete the
table.
data table
state of matter
at change
temperature at sea
level (°c)
temperature at high
elevation (°c)
liquid to gas
b. 12 °c
a. 0 °c
solid to liquid
c. 92 °c
c. 92 °c
liquid to solid
b. 12 °c
d. 100 °c
a. 0 °c
b. 12 °c
c. 92 °c
d. 100 °c
e. 106 °c
To solve this, we recall the phase change temperatures of water:
Liquid to Gas (Boiling):
- At sea level, water boils at \( 100^\circ \text{C} \), but at high elevation (lower atmospheric pressure), the boiling point decreases. However, the standard sea - level boiling point of water is \( 100^\circ \text{C} \), and if we consider typical values, for liquid to gas at sea level, the correct temperature is around \( 100^\circ \text{C} \) (option D), and at high elevation, it would be less, but from the given options, if we assume a general case (maybe a typo or simplified), but actually, the correct sea - level boiling point is \( 100^\circ \text{C} \).
Solid to Liquid (Melting):
- The melting point of ice (solid water to liquid water) at sea level is \( 0^\circ \text{C} \) (option A). At high elevation, the melting point of ice also remains approximately \( 0^\circ \text{C} \) (since melting point of pure water is not much affected by pressure for typical elevations), so for solid to liquid, at sea level and high elevation, it's \( 0^\circ \text{C} \) (option A).
Liquid to Solid (Freezing):
- The freezing point of water (liquid to solid) at sea level is \( 0^\circ \text{C} \) (option A). At high elevation, the freezing point of pure water is also approximately \( 0^\circ \text{C} \).
But let's re - evaluate with the given options:
- Liquid to Gas:
- At sea level, water boils at \( 100^\circ \text{C} \) (option D). At high elevation, if we take a value, but from the options, the closest for sea - level boiling is D (\( 100^\circ \text{C} \)).
- Solid to Liquid:
- The melting point of ice is \( 0^\circ \text{C} \) (option A) at sea level and high elevation (for pure water).
- Liquid to Solid:
- The freezing point of water is \( 0^\circ \text{C} \) (option A) at sea level and high elevation (for pure water).
If we correct the initial wrong crosses:
- Liquid to Gas (sea level): \( \boldsymbol{100^\circ \text{C}} \) (Option D)
- Liquid to Gas (high elevation): A value less than \( 100^\circ \text{C} \), but from the options, if we assume a mistake and the intended sea - level boiling is D.
- Solid to Liquid (sea level and high elevation): \( \boldsymbol{0^\circ \text{C}} \) (Option A)
- Liquid to Solid (sea level and high elevation): \( \boldsymbol{0^\circ \text{C}} \) (Option A)
Final Answers (correcting the table):
| State of matter at change | Temperature at sea level (\(^\circ \text{C}\)) | Temperature at high elevation (\(^\circ \text{C}\)) |
|---|
Let's start over with correct phase change knowledge:
- Melting (Solid to Liquid): Ice melts at \( 0^\circ \text{C} \) (at sea level and high elevation for pure water) → so solid to liquid: sea level = A (\( 0^\circ \text{C} \)), high elevation = A (\( 0^\circ \text{C} \)).
- Boiling (Liquid to Gas): At sea level, water boils at \( 100^\circ \text{C} \) (D), at high elevation (lower pressure) it boils at a lower temp…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we recall the phase change temperatures of water:
Liquid to Gas (Boiling):
- At sea level, water boils at \( 100^\circ \text{C} \), but at high elevation (lower atmospheric pressure), the boiling point decreases. However, the standard sea - level boiling point of water is \( 100^\circ \text{C} \), and if we consider typical values, for liquid to gas at sea level, the correct temperature is around \( 100^\circ \text{C} \) (option D), and at high elevation, it would be less, but from the given options, if we assume a general case (maybe a typo or simplified), but actually, the correct sea - level boiling point is \( 100^\circ \text{C} \).
Solid to Liquid (Melting):
- The melting point of ice (solid water to liquid water) at sea level is \( 0^\circ \text{C} \) (option A). At high elevation, the melting point of ice also remains approximately \( 0^\circ \text{C} \) (since melting point of pure water is not much affected by pressure for typical elevations), so for solid to liquid, at sea level and high elevation, it's \( 0^\circ \text{C} \) (option A).
Liquid to Solid (Freezing):
- The freezing point of water (liquid to solid) at sea level is \( 0^\circ \text{C} \) (option A). At high elevation, the freezing point of pure water is also approximately \( 0^\circ \text{C} \).
But let's re - evaluate with the given options:
- Liquid to Gas:
- At sea level, water boils at \( 100^\circ \text{C} \) (option D). At high elevation, if we take a value, but from the options, the closest for sea - level boiling is D (\( 100^\circ \text{C} \)).
- Solid to Liquid:
- The melting point of ice is \( 0^\circ \text{C} \) (option A) at sea level and high elevation (for pure water).
- Liquid to Solid:
- The freezing point of water is \( 0^\circ \text{C} \) (option A) at sea level and high elevation (for pure water).
If we correct the initial wrong crosses:
- Liquid to Gas (sea level): \( \boldsymbol{100^\circ \text{C}} \) (Option D)
- Liquid to Gas (high elevation): A value less than \( 100^\circ \text{C} \), but from the options, if we assume a mistake and the intended sea - level boiling is D.
- Solid to Liquid (sea level and high elevation): \( \boldsymbol{0^\circ \text{C}} \) (Option A)
- Liquid to Solid (sea level and high elevation): \( \boldsymbol{0^\circ \text{C}} \) (Option A)
Final Answers (correcting the table):
| State of matter at change | Temperature at sea level (\(^\circ \text{C}\)) | Temperature at high elevation (\(^\circ \text{C}\)) |
|---|
Let's start over with correct phase change knowledge:
- Melting (Solid to Liquid): Ice melts at \( 0^\circ \text{C} \) (at sea level and high elevation for pure water) → so solid to liquid: sea level = A (\( 0^\circ \text{C} \)), high elevation = A (\( 0^\circ \text{C} \)).
- Boiling (Liquid to Gas): At sea level, water boils at \( 100^\circ \text{C} \) (D), at high elevation (lower pressure) it boils at a lower temperature, e.g., \( 92^\circ \text{C} \) (C).
- Freezing (Liquid to Solid): Water freezes at \( 0^\circ \text{C} \) (at sea level and high elevation for pure water) → liquid to solid: sea level = A (\( 0^\circ \text{C} \)), high elevation = A (\( 0^\circ \text{C} \)).
So the correct assignments are:
- Liquid to gas (sea level): D (\( 100^\circ \text{C} \))
- Liquid to gas (high elevation): C (\( 92^\circ \text{C} \))
- Solid to liquid (sea level): A (\( 0^\circ \text{C} \))
- Solid to liquid (high elevation): A (\( 0^\circ \text{C} \))
- Liquid to solid (sea level): A (\( 0^\circ \text{C} \))
- Liquid to solid (high elevation): A (\( 0^\circ \text{C} \))
If we consider the initial wrong marks (the red x's), we need to replace them with the correct values:
- For Liquid to Gas (sea level): Replace the wrong B (\( 12^\circ \text{C} \)) with D (\( 100^\circ \text{C} \)).
- For Liquid to Gas (high elevation): Replace the wrong A (\( 0^\circ \text{C} \)) with C (\( 92^\circ \text{C} \)).
- For Solid to Liquid (sea level): Replace the wrong C (\( 92^\circ \text{C} \)) with A (\( 0^\circ \text{C} \)).
- For Solid to Liquid (high elevation): Replace the wrong C (\( 92^\circ \text{C} \)) with A (\( 0^\circ \text{C} \)).
- For Liquid to Solid (sea level): Replace the wrong B (\( 12^\circ \text{C} \)) with A (\( 0^\circ \text{C} \)).
- For Liquid to Solid (high elevation): Replace the wrong D (\( 100^\circ \text{C} \)) with A (\( 0^\circ \text{C} \)).
Final Answer (correcting each cell):
| State of matter at change | Temperature at sea level (\(^\circ \text{C}\)) | Temperature at high elevation (\(^\circ \text{C}\)) |
|---|---|---|
| Solid to liquid | A (\( 0^\circ \text{C} \)) | A (\( 0^\circ \text{C} \)) |
| Liquid to solid | A (\( 0^\circ \text{C} \)) | A (\( 0^\circ \text{C} \)) |
So the correct temperatures to drag:
- Liquid to gas (sea level): D (\( 100^\circ \text{C} \))
- Liquid to gas (high elevation): C (\( 92^\circ \text{C} \))
- Solid to liquid (sea level): A (\( 0^\circ \text{C} \))
- Solid to liquid (high elevation): A (\( 0^\circ \text{C} \))
- Liquid to solid (sea level): A (\( 0^\circ \text{C} \))
- Liquid to solid (high elevation): A (\( 0^\circ \text{C} \))
If we just give the answers for each row:
- Liquid to Gas:
- Sea level: D. \( 100^\circ \text{C} \)
- High elevation: C. \( 92^\circ \text{C} \)
- Solid to Liquid:
- Sea level: A. \( 0^\circ \text{C} \)
- High elevation: A. \( 0^\circ \text{C} \)
- Liquid to Solid:
- Sea level: A. \( 0^\circ \text{C} \)
- High elevation: A. \( 0^\circ \text{C} \)
The main correct temperatures for each phase change (from the options) are:
- Solid to Liquid: A (\( 0^\circ \text{C} \))
- Liquid to Solid: A (\( 0^\circ \text{C} \))
- Liquid to Gas (sea level): D (\( 100^\circ \text{C} \)), (high elevation): C (\( 92^\circ \text{C} \))
So the answer for each cell (correcting the x's) is:
- Liquid to Gas (sea level): D (\( 100^\circ \text{C} \))
- Liquid to Gas (high elevation): C (\( 92^\circ \text{C} \))
- Solid to Liquid (sea level): A (\( 0^\circ \text{C} \))
- Solid to Liquid (high elevation): A (\( 0^\circ \text{C} \))
- Liquid to Solid (sea level): A (\( 0^\circ \text{C} \))
- Liquid to Solid (high elevation): A (\( 0^\circ \text{C} \))
If we have to choose one per phase change (assuming the table is for a general case):
- Liquid to Gas: D (\( 100^\circ \text{C} \)) (sea level)
- Solid to Liquid: A (\( 0^\circ \text{C} \))
- Liquid to Solid: A (\( 0^\circ \text{C} \))
So the final answers (matching the options) are:
- Liquid to Gas (sea level): D. \( 100^\circ \text{C} \)
- Solid to Liquid: A. \( 0^\circ \text{C} \)
- Liquid to Solid: A. \( 0^\circ \text{C} \)
- Liquid to Gas (high elevation): C. \( 92^\circ \text{C} \)
(Note: The initial x's were wrong, and we corrected them based on the phase change temperatures of water.)