QUESTION IMAGE
Question
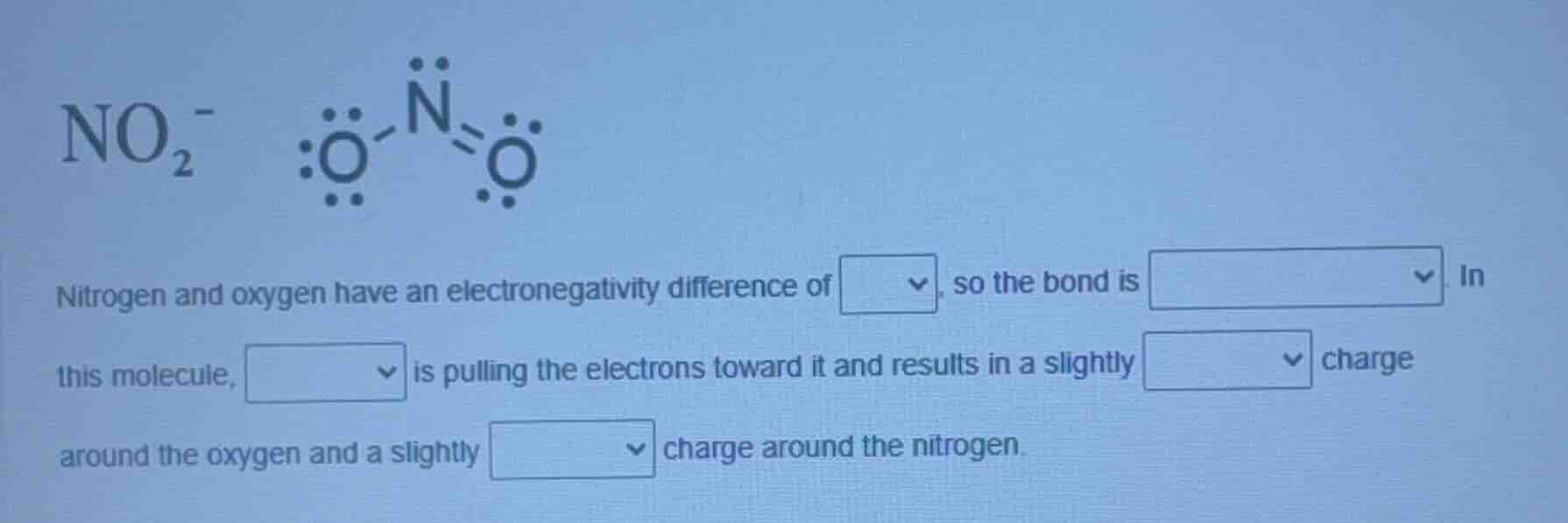
no₂⁻
nitrogen and oxygen have an electronegativity difference of △, so the bond is △. in this molecule, △ is pulling the electrons toward it and results in a slightly △ charge around the oxygen and a slightly △ charge around the nitrogen.
Step1: Determine electronegativity values
Electronegativity of N is ~3.0, O is ~3.5. Difference: \( 3.5 - 3.0 = 0.5 \).
Step2: Classify bond type
Electronegativity difference \( \leq 0.5 \) (or small) indicates polar covalent (since 0.5 is between nonpolar (≤0.4) and polar covalent (0.5 - 1.6)).
Step3: Identify electron puller
Oxygen (O) is more electronegative than N, so O pulls electrons.
Step4: Determine charges
More electronegative atom (O) gets slightly negative charge, less electronegative (N) gets slightly positive charge.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Electronegativity difference: \( 0.5 \); Bond type: polar covalent; Electron puller: oxygen (O); Oxygen charge: negative; Nitrogen charge: positive