QUESTION IMAGE
Question
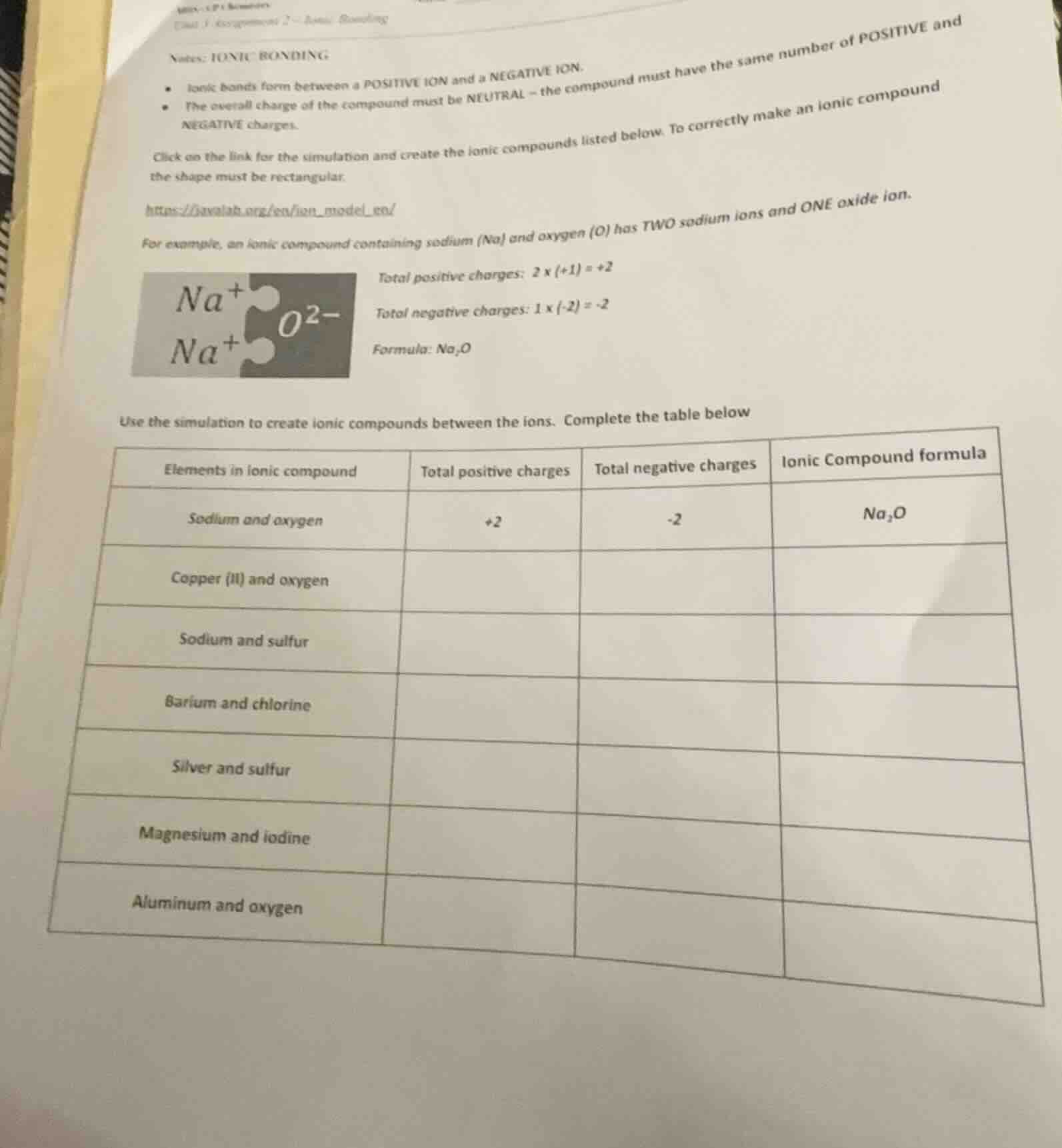
notes: ionic bonding
- ionic bonds form between a positive ion and a negative ion.
- the overall charge of the compound must be neutral - the compound must have the same number of positive and negative charges.
click on the link for the simulation and create the ionic compounds listed below. to correctly make an ionic compound the shape must be rectangular.
https://phet.colorado.edu/en/simulation/ion - model - mc/
for example, an ionic compound containing sodium (na) and oxygen (o) has two sodium ions and one oxide ion.
na⁺ o²⁻
total positive charges: 2×(+1) = +2
total negative charges: 1×(-2) = -2
formula: na₂o
use the simulation to create ionic compounds between the ions. complete the table below
| elements in ionic compound | total positive charges | total negative charges | ionic compound formula |
|---|---|---|---|
| copper (ii) and oxygen | |||
| sodium and sulfur | |||
| barium and chlorine | |||
| silver and sulfur | |||
| magnesium and iodine | |||
| aluminum and oxygen |
Step1: Identify ion charges
Copper (II): $Cu^{2+}$ (+2), Oxygen: $O^{2-}$ (-2)
Step2: Balance total charges
Total positive: $1 \times (+2) = +2$
Total negative: $1 \times (-2) = -2$
Formula: $CuO$
Step3: Identify ion charges
Sodium: $Na^{+}$ (+1), Sulfur: $S^{2-}$ (-2)
Step4: Balance total charges
Total positive: $2 \times (+1) = +2$
Total negative: $1 \times (-2) = -2$
Formula: $Na_2S$
Step5: Identify ion charges
Barium: $Ba^{2+}$ (+2), Chlorine: $Cl^{-}$ (-1)
Step6: Balance total charges
Total positive: $1 \times (+2) = +2$
Total negative: $2 \times (-1) = -2$
Formula: $BaCl_2$
Step7: Identify ion charges
Silver: $Ag^{+}$ (+1), Sulfur: $S^{2-}$ (-2)
Step8: Balance total charges
Total positive: $2 \times (+1) = +2$
Total negative: $1 \times (-2) = -2$
Formula: $Ag_2S$
Step9: Identify ion charges
Magnesium: $Mg^{2+}$ (+2), Iodine: $I^{-}$ (-1)
Step10: Balance total charges
Total positive: $1 \times (+2) = +2$
Total negative: $2 \times (-1) = -2$
Formula: $MgI_2$
Step11: Identify ion charges
Aluminum: $Al^{3+}$ (+3), Oxygen: $O^{2-}$ (-2)
Step12: Balance total charges
Total positive: $2 \times (+3) = +6$
Total negative: $3 \times (-2) = -6$
Formula: $Al_2O_3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Elements in ionic compound | Total positive charges | Total negative charges | Ionic Compound formula |
|---|---|---|---|
| Copper (II) and oxygen | +2 | -2 | $CuO$ |
| Sodium and sulfur | +2 | -2 | $Na_2S$ |
| Barium and chlorine | +2 | -2 | $BaCl_2$ |
| Silver and sulfur | +2 | -2 | $Ag_2S$ |
| Magnesium and iodine | +2 | -2 | $MgI_2$ |
| Aluminum and oxygen | +6 | -6 | $Al_2O_3$ |