QUESTION IMAGE
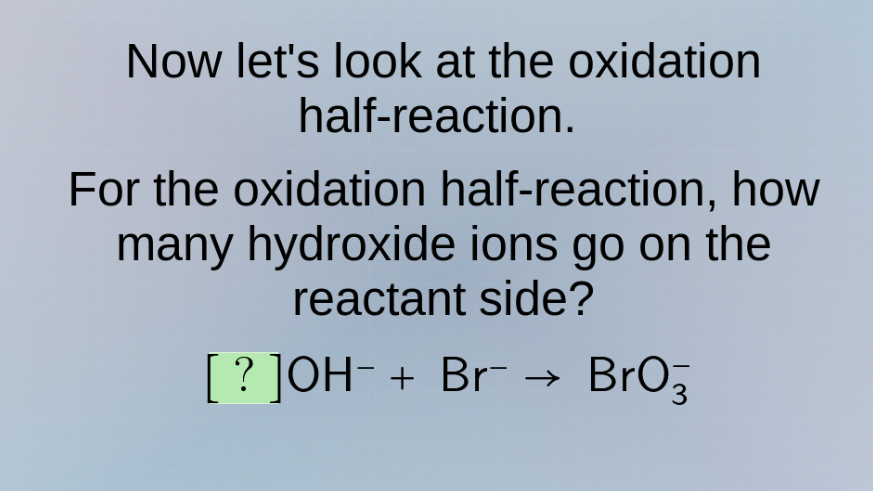
Question
now lets look at the oxidation half - reaction. for the oxidation half - reaction, how many hydroxide ions go on the reactant side? ?oh⁻ + br⁻ → bro₃⁻
Step1: Balance Br atoms
The Br atoms are already balanced (1 Br on each side).
Step2: Balance O atoms
On the product side, there are 3 O atoms in \( \text{BrO}_3^- \). To balance O, we add \( \text{H}_2\text{O} \) on the reactant side? Wait, no, in basic solution, we use \( \text{OH}^- \) and \( \text{H}_2\text{O} \). Wait, let's do it properly.
First, let's write the unbalanced half-reaction: \( \text{Br}^-
ightarrow \text{BrO}_3^- \) (oxidation, since Br goes from -1 to +5, loss of electrons).
Balance O: Add 3 \( \text{H}_2\text{O} \) on the left? No, in basic solution, we balance O by adding \( \text{OH}^- \) and \( \text{H}_2\text{O} \). Wait, the correct approach for basic solution:
- Balance Br: \( \text{Br}^-
ightarrow \text{BrO}_3^- \) (already 1 Br each)
- Balance O: Add 3 \( \text{H}_2\text{O} \) on the left? No, wait, in basic solution, to balance O, we add \( \text{OH}^- \) to the side with less O and \( \text{H}_2\text{O} \) to the other. Wait, the product has 3 O, so we need 3 O on the left. So add 3 \( \text{H}_2\text{O} \) on the left? No, that would add H. Wait, maybe better to balance in acidic first then convert to basic.
In acidic solution:
- Balance Br: \( \text{Br}^-
ightarrow \text{BrO}_3^- \)
- Balance O: Add 3 \( \text{H}_2\text{O} \) on left: \( 3\text{H}_2\text{O} + \text{Br}^-
ightarrow \text{BrO}_3^- \)
- Balance H: Add 6 \( \text{H}^+ \) on right: \( 3\text{H}_2\text{O} + \text{Br}^-
ightarrow \text{BrO}_3^- + 6\text{H}^+ \)
- Balance charge: Left charge: -1, Right charge: -1 + 6(+1) = +5. So add 6 \( e^- \) on right: \( 3\text{H}_2\text{O} + \text{Br}^-
ightarrow \text{BrO}_3^- + 6\text{H}^+ + 6e^- \)
Now convert to basic by adding 6 \( \text{OH}^- \) to both sides:
Left: \( 3\text{H}_2\text{O} + \text{Br}^- + 6\text{OH}^- \)
Right: \( \text{BrO}_3^- + 6\text{H}^+ + 6e^- + 6\text{OH}^- \)
Combine \( \text{H}^+ \) and \( \text{OH}^- \) to form \( \text{H}_2\text{O} \):
Right: \( \text{BrO}_3^- + 6\text{H}_2\text{O} + 6e^- \)
Now simplify \( \text{H}_2\text{O} \):
Left has 3 \( \text{H}_2\text{O} \), right has 6 \( \text{H}_2\text{O} \). Subtract 3 \( \text{H}_2\text{O} \) from both sides:
Left: \( \text{Br}^- + 6\text{OH}^- \)
Right: \( \text{BrO}_3^- + 3\text{H}_2\text{O} + 6e^- \)
Wait, but the original equation given is \( \text{OH}^- + \text{Br}^-
ightarrow \text{BrO}_3^- \) (missing some terms, like \( \text{H}_2\text{O} \) and electrons, but the question is about \( \text{OH}^- \) on reactant side. Wait, maybe the equation is missing \( \text{H}_2\text{O} \) and electrons, but let's check the O balance.
Wait, the given equation is \( [?]\text{OH}^- + \text{Br}^-
ightarrow \text{BrO}_3^- \). Let's balance O: product has 3 O, so reactant needs 3 O from \( \text{OH}^- \)? No, \( \text{OH}^- \) has 1 O each, so 3 \( \text{OH}^- \) would give 3 O, but then we have H. Wait, maybe the correct balancing for basic solution:
The correct half-reaction (oxidation) in basic solution:
- Balance Br: \( \text{Br}^-
ightarrow \text{BrO}_3^- \)
- Balance O: Add 3 \( \text{H}_2\text{O} \) to the left (wait, no, in basic, to balance O, we add \( \text{OH}^- \) to the side with less O. Wait, product has 3 O, so we need 3 O on left. So add 3 \( \text{OH}^- \) to left? No, that would add H. Wait, I think I made a mistake earlier. Let's do it properly for basic solution:
Steps for balancing oxidation half-reaction in basic solution:
a. Balance Br: \( \text{Br}^-
ightarrow \text{BrO}_3^- \) (1 Br each)
b. Balance O: Add 3 \( \text{H}_2\text{O} \) to the left (to provide O): \( 3\text{H}_2\text…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6