QUESTION IMAGE
Question
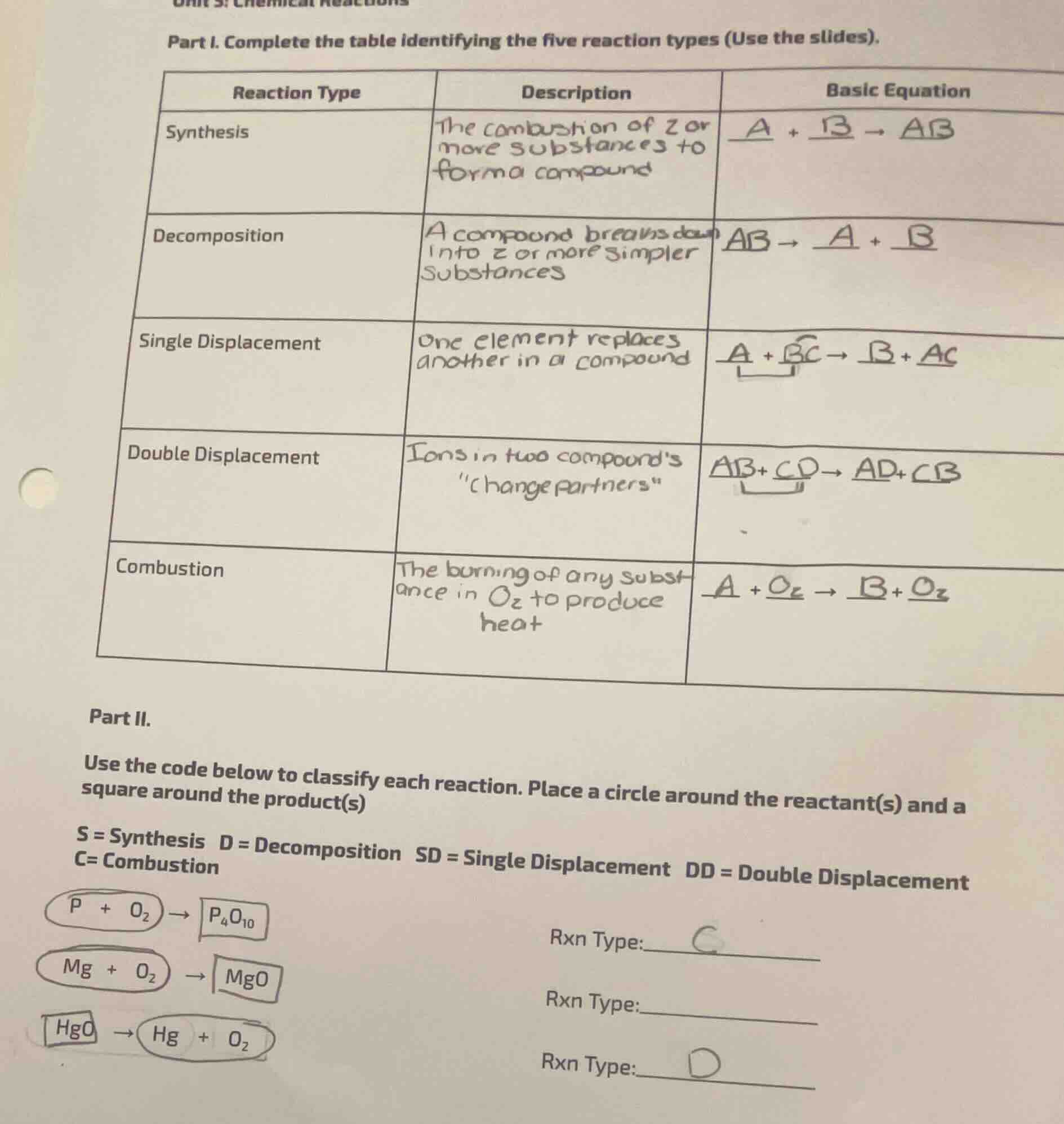
part i. complete the table identifying the five reaction types (use the slides).
| reaction type | description | basic equation |
|---|
ightarrow \underline{ab}$ |
| decomposition | a compound breaks down into 2 or more simpler substances | $\underline{ab} \ |
ightarrow \underline{a} + \underline{b}$ |
| single displacement | one element replaces another in a compound | $\underline{a} + \underline{bc} \ |
ightarrow \underline{b} + \underline{ac}$ |
| double displacement | ions in two compounds \change partners\ | $\underline{ab} + \underline{cd} \ |
ightarrow \underline{ad} + \underline{cb}$ |
| combustion | the burning of any substance in $o_2$ to produce heat | $\underline{a} + \underline{o_2} \ |
ightarrow \underline{b} + \underline{o_2}$ |
part ii.
use the code below to classify each reaction. place a circle around the reactant(s) and a square around the product(s)
$s = $ synthesis $d = $ decomposition $sd = $ single displacement $dd = $ double displacement $c = $ combustion
$\boldsymbol{\bigcirc (p + o_2) \
ightarrow \square (p_4o_{10})}$ rxn type: $\underline{c}$
$\boldsymbol{\bigcirc (mg + o_2) \
ightarrow \square (mgo)}$ rxn type: $\underline{\quad}$
$\boldsymbol{\square (hgo) \
ightarrow \bigcirc (hg + o_2)}$ rxn type: $\underline{d}$
Part I: Correcting the Table (if needed, but here we focus on Part II classification)
Reaction 1: $\boldsymbol{\text{P} + \text{O}_2
ightarrow \text{P}_4\text{O}_{10}}$
Step1: Analyze Reaction Type
Synthesis reaction is when 2 or more substances combine to form a compound. Combustion is a type of synthesis with $\text{O}_2$, but also, the general synthesis (and combustion here) fits. Wait, the code: S = Synthesis, C = Combustion. But $\text{P} + \text{O}_2$ forming $\text{P}_4\text{O}_{10}$ is both combustion (burning in $\text{O}_2$) and synthesis (multiple reactants forming one product). But let's check the code: the first reaction's Rxn Type was marked C, but also, synthesis: $\text{A} + \text{B}
ightarrow \text{AB}$ (here multiple P and $\text{O}_2$ form $\text{P}_4\text{O}_{10}$). Wait, maybe the initial marking was wrong? Wait, no—combustion is a type of reaction where a substance reacts with $\text{O}_2$ to produce heat, and often is a synthesis. But let's re - evaluate:
The reaction $\text{P} + \text{O}_2
ightarrow \text{P}_4\text{O}_{10}$: reactants are P and $\text{O}_2$ (two substances), product is $\text{P}_4\text{O}_{10}$ (one compound). So synthesis (S) as per the basic equation $\text{A} + \text{B}
ightarrow \text{AB}$ (here A = P, B = $\text{O}_2$, AB = $\text{P}_4\text{O}_{10}$). Also, it's combustion (C) as it's burning in $\text{O}_2$. But maybe the intended is S? Wait, no—the first reaction's initial marking was C, but let's check the second reaction.
Reaction 2: $\boldsymbol{\text{Mg} + \text{O}_2
ightarrow \text{MgO}}$
Step1: Analyze Reaction Type
Reactants: Mg and $\text{O}_2$ (two substances), product: MgO (one compound). This fits the synthesis reaction (S) equation $\text{A} + \text{B}
ightarrow \text{AB}$ (A = Mg, B = $\text{O}_2$, AB = MgO). It's also combustion (burning in $\text{O}_2$), but the synthesis type is also applicable. But let's check the code: S = Synthesis.
Reaction 3: $\boldsymbol{\text{HgO}
ightarrow \text{Hg} + \text{O}_2}$
Step1: Analyze Reaction Type
Reactant: HgO (one compound), products: Hg and $\text{O}_2$ (two simpler substances). This fits the decomposition reaction (D) equation $\text{AB}
ightarrow \text{A} + \text{B}$ (AB = HgO, A = Hg, B = $\text{O}_2$). The initial marking D is correct.
Part II: Correcting the Reaction Types
- For $\text{P} + \text{O}_2
ightarrow \text{P}_4\text{O}_{10}$:
- It can be classified as S (Synthesis) (since multiple reactants form one product) or C (Combustion) (since it reacts with $\text{O}_2$ to produce heat). But if we follow the basic equation for synthesis ($\text{A} + \text{B}
ightarrow \text{AB}$), this is synthesis. However, the initial marking was C, which is also correct as combustion is a special case of synthesis with $\text{O}_2$. But maybe the intended answer for the first reaction (after correction) considering the table's synthesis description: "the combination of 2 or more substances to form a compound"—so S. But the first reaction was marked C, maybe a mistake. Let's proceed with the correct classification based on the reaction types:
Reaction 1 ( $\text{P} + \text{O}_2
ightarrow \text{P}_4\text{O}_{10}$):
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s for Part II:
- $\text{P} + \text{O}_2
ightarrow \text{P}_4\text{O}_{10}$: Rxn Type: $\boldsymbol{\text{S (or C)}}$ (if we take synthesis: S, if combustion: C. But based on the table's synthesis description "combination of 2 or more substances to form a compound", S is also correct. However, the initial marking was C, maybe the answer is C)
- $\text{Mg} + \text{O}_2
ightarrow \text{MgO}$: Rxn Type: $\boldsymbol{\text{S}}$
- $\text{HgO}
ightarrow \text{Hg} + \text{O}_2$: Rxn Type: $\boldsymbol{\text{D}}$
(Note: There might be some confusion between combustion and synthesis as combustion is a type of synthesis with $\text{O}_2$. The first reaction can be classified as both S and C, but based on the code and the table, if we follow the initial marking's logic for the first reaction as C, then:
- Rxn Type: C
- Rxn Type: S (or C)
- Rxn Type: D
But the most accurate for the second reaction (Mg + O₂ → MgO) is S (synthesis) as it fits the $\text{A} + \text{B}
ightarrow \text{AB}$ pattern.)