QUESTION IMAGE
Question
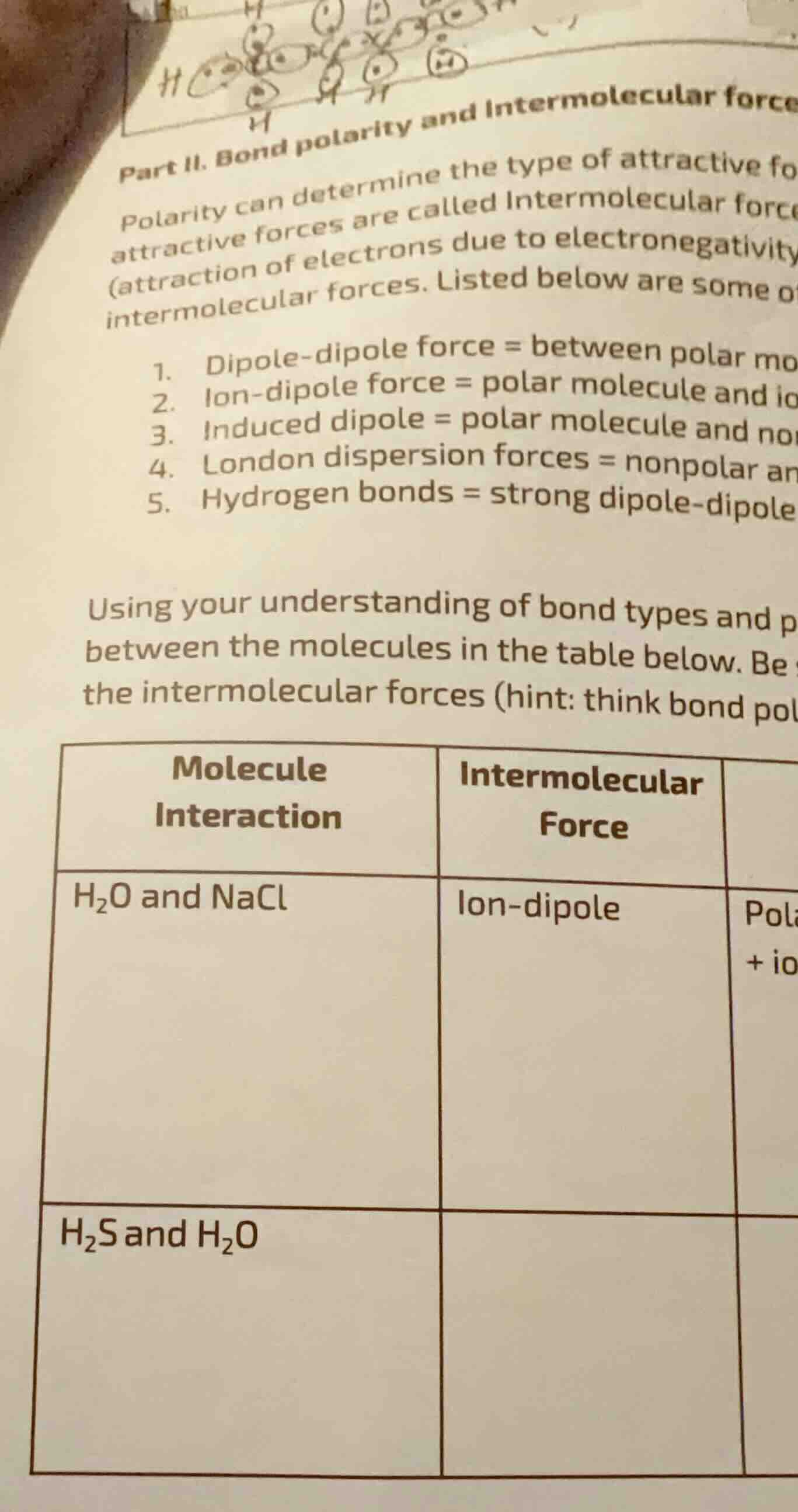
part ii. bond polarity and intermolecular force
polarity can determine the type of attractive fo
attractive forces are called intermolecular force
(attraction of electrons due to electronegativity
intermolecular forces. listed below are some o
- dipole-dipole force = between polar mo
- ion-dipole force = polar molecule and io
- induced dipole = polar molecule and no
- london dispersion forces = nonpolar an
- hydrogen bonds = strong dipole-dipole
using your understanding of bond types and p
between the molecules in the table below. be
the intermolecular forces (hint: think bond pol
| molecule interaction | intermolecular force |
|---|---|
| h₂s and h₂o |
Step1: Analyze the polarity of molecules
Both \( \ce{H2S} \) and \( \ce{H2O} \) are polar molecules. \( \ce{H2O} \) has a bent molecular geometry with a significant dipole moment due to the electronegativity difference between O and H. \( \ce{H2S} \) also has a bent geometry (similar to \( \ce{H2O} \) in terms of molecular shape, though S is less electronegative than O), and it is a polar molecule because of the electronegativity difference between S and H.
Step2: Determine the intermolecular force
Since both molecules are polar, the intermolecular force between them is dipole - dipole interaction. Dipole - dipole forces occur between polar molecules, where the positive end of one dipole is attracted to the negative end of another dipole.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For the interaction between \( \ce{H2S} \) and \( \ce{H2O} \), the intermolecular force is Dipole - dipole force.