QUESTION IMAGE
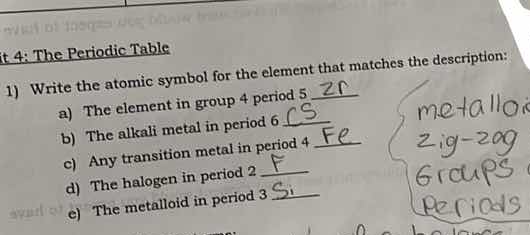
Question
t 4: the periodic table
- write the atomic symbol for the element that matches the description:
a) the element in group 4 period 5 zr
b) the alkali metal in period 6 cs
c) any transition metal in period 4 fe
d) the halogen in period 2
e) the metalloid in period 3 si
To solve these problems, we use knowledge of the periodic table, including group and period numbering, and the properties of different element categories (alkali metals, transition metals, halogens, metalloids).
Part (a)
Group 4, period 5: Elements in group 4 are transition metals with 4 valence electrons (in the d - block for periods 4 and above). In period 5, group 4, the element is zirconium. The atomic symbol for zirconium is $\boldsymbol{\ce{Zr}}$.
Part (b)
Alkali metals are in group 1. In period 6, the alkali metal is cesium. The atomic symbol for cesium is $\boldsymbol{\ce{Cs}}$.
Part (c)
Transition metals are in the d - block (groups 3 - 12). In period 4, examples include iron ($\ce{Fe}$), copper ($\ce{Cu}$), zinc ($\ce{Zn}$), etc. A common transition metal in period 4 is iron, with atomic symbol $\boldsymbol{\ce{Fe}}$.
Part (d)
Halogens are in group 17. In period 2, the halogen is fluorine. The atomic symbol for fluorine is $\boldsymbol{\ce{F}}$.
Part (e)
Metalloids have properties of both metals and non - metals. In period 3, the metalloid is silicon. The atomic symbol for silicon is $\boldsymbol{\ce{Si}}$.
Final Answers
a) $\boldsymbol{\ce{Zr}}$
b) $\boldsymbol{\ce{Cs}}$
c) $\boldsymbol{\ce{Fe}}$ (or other valid transition metal in period 4)
d) $\boldsymbol{\ce{F}}$
e) $\boldsymbol{\ce{Si}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve these problems, we use knowledge of the periodic table, including group and period numbering, and the properties of different element categories (alkali metals, transition metals, halogens, metalloids).
Part (a)
Group 4, period 5: Elements in group 4 are transition metals with 4 valence electrons (in the d - block for periods 4 and above). In period 5, group 4, the element is zirconium. The atomic symbol for zirconium is $\boldsymbol{\ce{Zr}}$.
Part (b)
Alkali metals are in group 1. In period 6, the alkali metal is cesium. The atomic symbol for cesium is $\boldsymbol{\ce{Cs}}$.
Part (c)
Transition metals are in the d - block (groups 3 - 12). In period 4, examples include iron ($\ce{Fe}$), copper ($\ce{Cu}$), zinc ($\ce{Zn}$), etc. A common transition metal in period 4 is iron, with atomic symbol $\boldsymbol{\ce{Fe}}$.
Part (d)
Halogens are in group 17. In period 2, the halogen is fluorine. The atomic symbol for fluorine is $\boldsymbol{\ce{F}}$.
Part (e)
Metalloids have properties of both metals and non - metals. In period 3, the metalloid is silicon. The atomic symbol for silicon is $\boldsymbol{\ce{Si}}$.
Final Answers
a) $\boldsymbol{\ce{Zr}}$
b) $\boldsymbol{\ce{Cs}}$
c) $\boldsymbol{\ce{Fe}}$ (or other valid transition metal in period 4)
d) $\boldsymbol{\ce{F}}$
e) $\boldsymbol{\ce{Si}}$