QUESTION IMAGE
Question
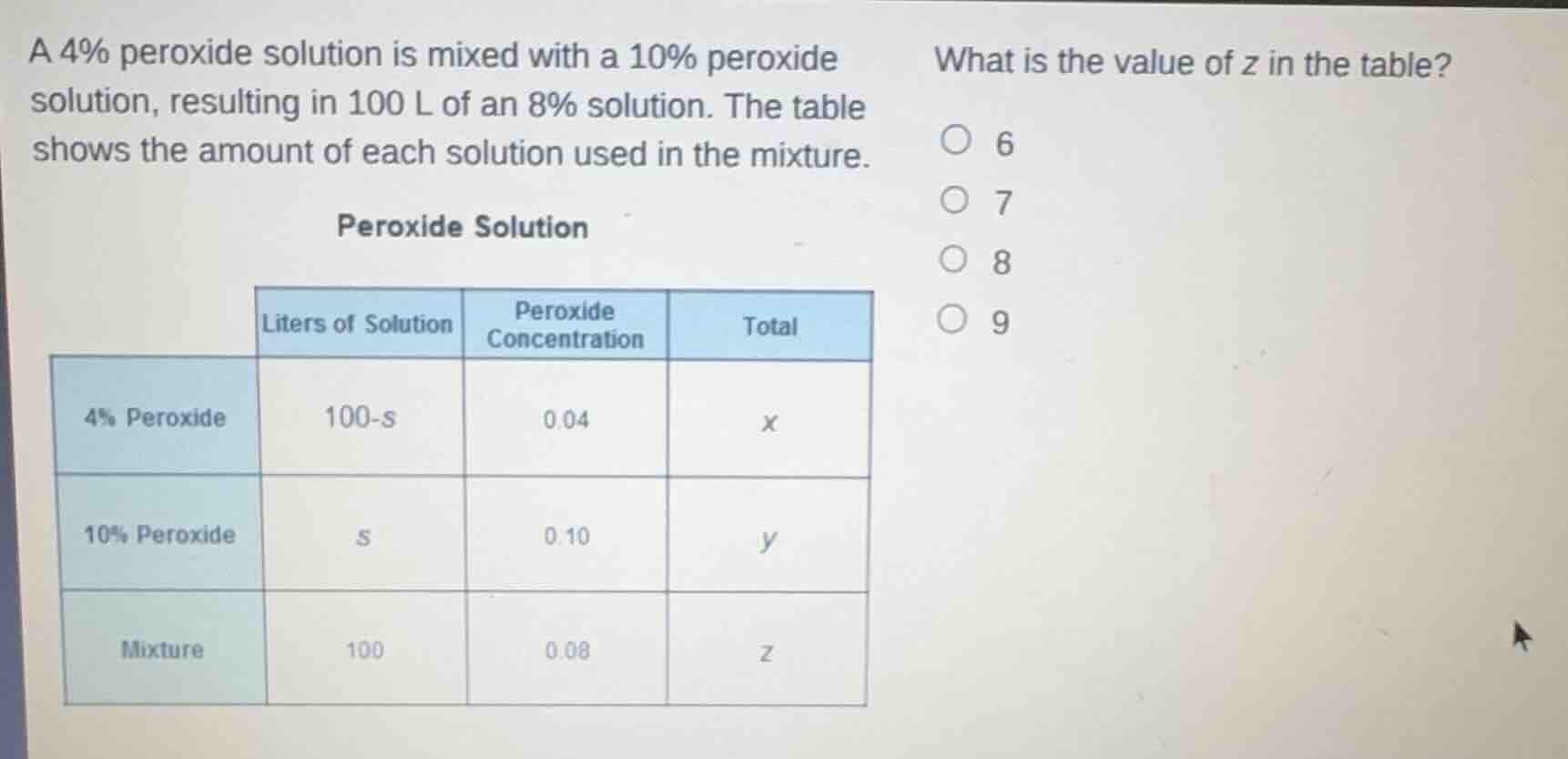
a 4% peroxide solution is mixed with a 10% peroxide solution, resulting in 100 l of an 8% solution. the table shows the amount of each solution used in the mixture.
peroxide solution
| liters of solution | peroxide concentration | total | |
|---|---|---|---|
| 10% peroxide | s | 0.10 | y |
| mixture | 100 | 0.08 | z |
what is the value of z in the table?
○ 6
○ 7
○ 8
○ 9
Step1: Recall the formula for mixture problems
The total amount of peroxide in the mixture is the sum of the peroxide from each solution. For the mixture, the total peroxide \( z \) can be calculated by multiplying the volume of the mixture (100 L) by its concentration (0.08). So we use the formula \( \text{Total Peroxide} = \text{Volume} \times \text{Concentration} \).
Step2: Calculate z
Substitute the values into the formula: \( z = 100\times0.08 \).
Calculating this, we get \( z = 8 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
8