QUESTION IMAGE
Question
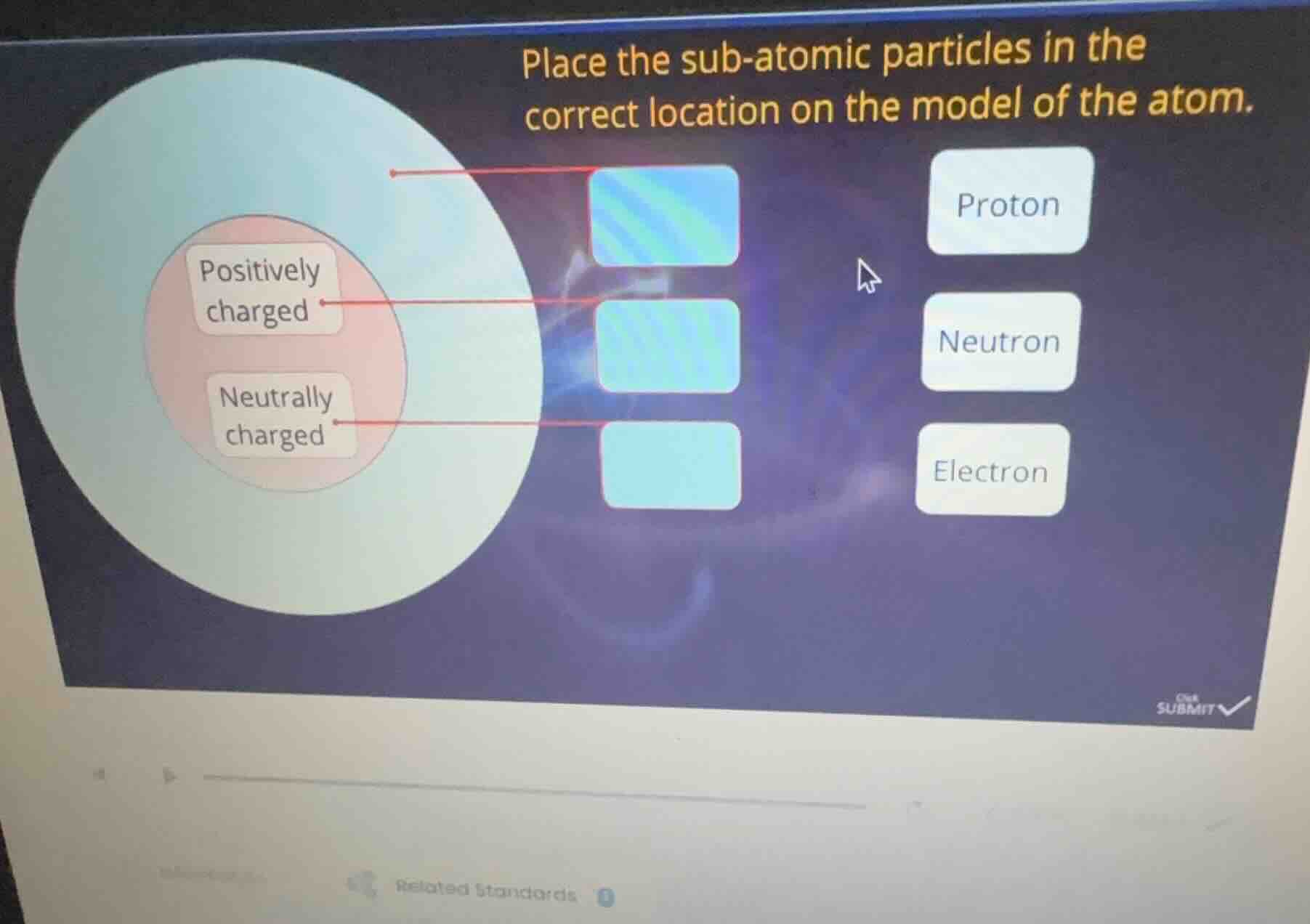
place the sub-atomic particles in the correct location on the model of the atom. positively charged neutrally charged proton neutron electron
Brief Explanations
- The outermost region (the light - blue area around the nucleus) is where electrons are located. Electrons are negatively charged and orbit the nucleus. So the top - most blue box (the one with the red arrow pointing to the outer region) should be filled with "Electron".
- The "Positively charged" region in the nucleus (the pink area) is where protons are located. Protons have a positive charge and are part of the atomic nucleus. So the middle blue box (with the red arrow pointing to the "Positively charged" part of the nucleus) should be filled with "Proton".
- The "Neutrally charged" region in the nucleus is where neutrons are located. Neutrons have no charge (neutral) and are also part of the atomic nucleus. So the bottom blue box (with the red arrow pointing to the "Neutrally charged" part of the nucleus) should be filled with "Neutron".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Top blue box (outer region): Electron
- Middle blue box (positively charged nucleus region): Proton
- Bottom blue box (neutrally charged nucleus region): Neutron