QUESTION IMAGE
Question
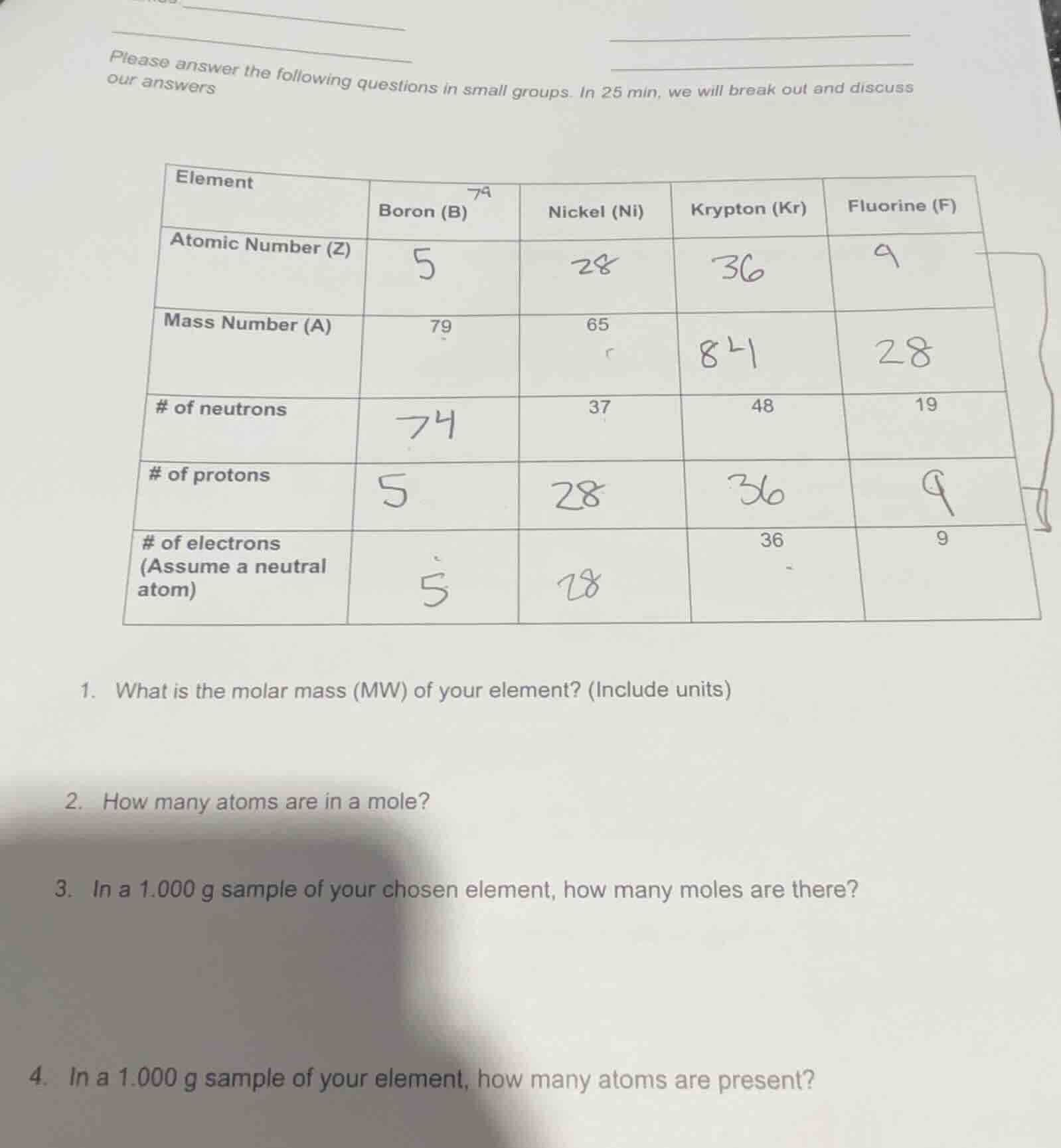
please answer the following questions in small groups. in 25 min, we will break out and discuss our answers
element | boron (b) | nickel (ni) | krypton (kr) | fluorine (f)
--- | --- | --- | --- | ---
atomic number (z) | 5 | 28 | 36 | 9
mass number (a) | 79 | 65 | 84 | 28
of neutrons | 74 | 37 | 48 | 19
of protons | 5 | 28 | 36 | 9
of electrons (assume a neutral atom) | 5 | 28 | 36 | 9
- what is the molar mass (mw) of your element? (include units)
- how many atoms are in a mole?
- in a 1.000 g sample of your chosen element, how many moles are there?
- in a 1.000 g sample of your element, how many atoms are present?
We will use Fluorine (F) as the chosen element for all calculations.
Step1: Identify molar mass
Molar mass equals mass number for the isotope, with units g/mol.
$\text{Molar Mass (F)} = 28\ \text{g/mol}$
Step2: State Avogadro's number
This is the number of atoms per mole.
$\text{Avogadro's Number} = 6.022 \times 10^{23}\ \text{atoms/mol}$
Step3: Calculate moles in 1.000 g
Use $\text{Moles} = \frac{\text{Mass}}{\text{Molar Mass}}$
$\text{Moles of F} = \frac{1.000\ \text{g}}{28\ \text{g/mol}} = 0.03571\ \text{mol}$
Step4: Calculate atoms in 1.000 g
Multiply moles by Avogadro's number.
$\text{Atoms of F} = 0.03571\ \text{mol} \times 6.022 \times 10^{23}\ \text{atoms/mol} = 2.151 \times 10^{22}\ \text{atoms}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $28\ \text{g/mol}$
- $6.022 \times 10^{23}\ \text{atoms}$
- $0.0357\ \text{mol}$ (rounded to 4 significant figures)
- $2.15 \times 10^{22}\ \text{atoms}$ (rounded to 3 significant figures)