QUESTION IMAGE
Question
(1 point)
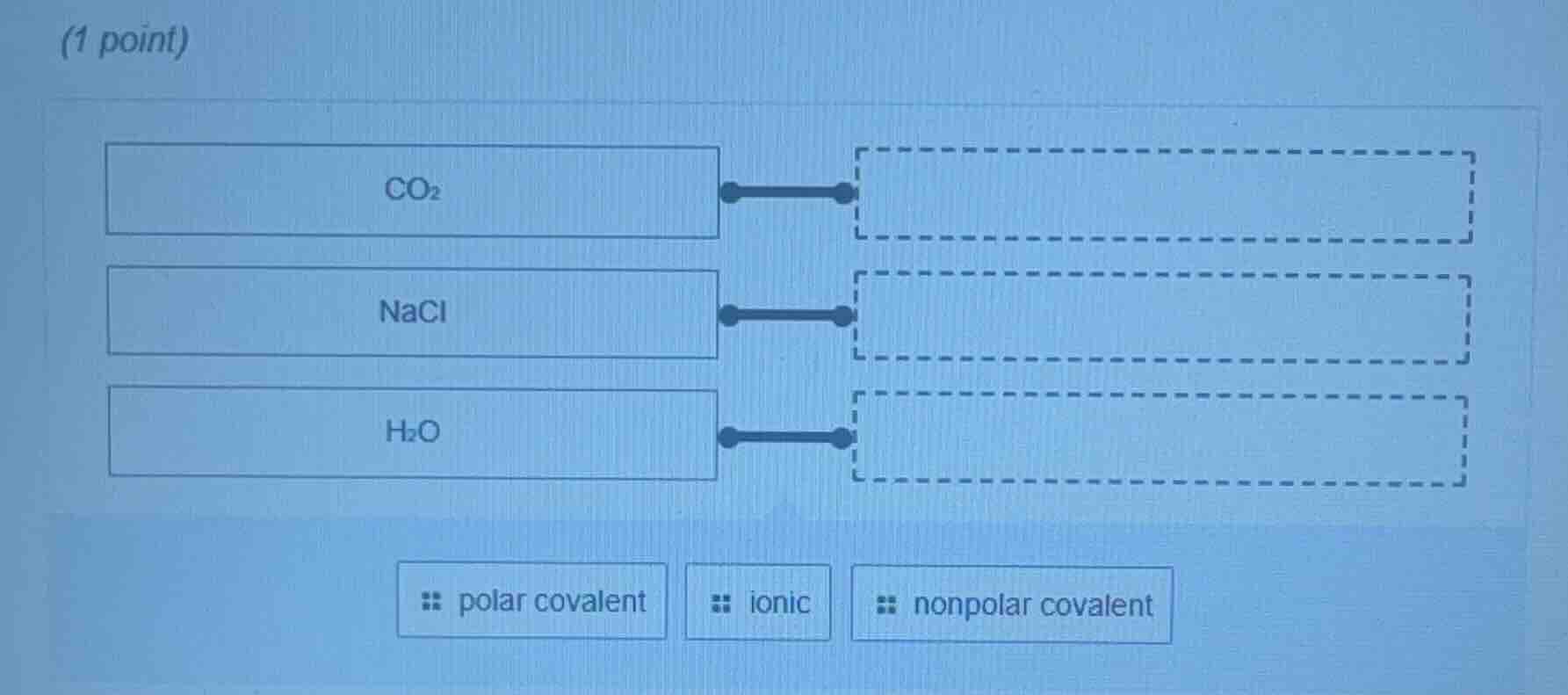
co₂
nacl
h₂o
polar covalent ionic nonpolar covalent
Step1: Analyze CO₂
Carbon dioxide (\(CO_2\)) has a linear structure with two \(C = O\) bonds. The electronegativity difference between \(C\) and \(O\) creates polar bonds, but the symmetric linear arrangement makes the molecule's dipole moments cancel out. So \(CO_2\) has nonpolar covalent bonds.
Step2: Analyze NaCl
Sodium chloride (\(NaCl\)) is formed by the transfer of an electron from \(Na\) (a metal) to \(Cl\) (a non - metal). This results in the formation of \(Na^+\) and \(Cl^-\) ions, and the bond between them is an ionic bond.
Step3: Analyze H₂O
Water (\(H_2O\)) has a bent structure. The \(O - H\) bonds are polar (due to the electronegativity difference between \(O\) and \(H\)), and the bent structure means the dipole moments do not cancel out. So \(H_2O\) has polar covalent bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(CO_2\) - nonpolar covalent; \(NaCl\) - ionic; \(H_2O\) - polar covalent