QUESTION IMAGE
Question
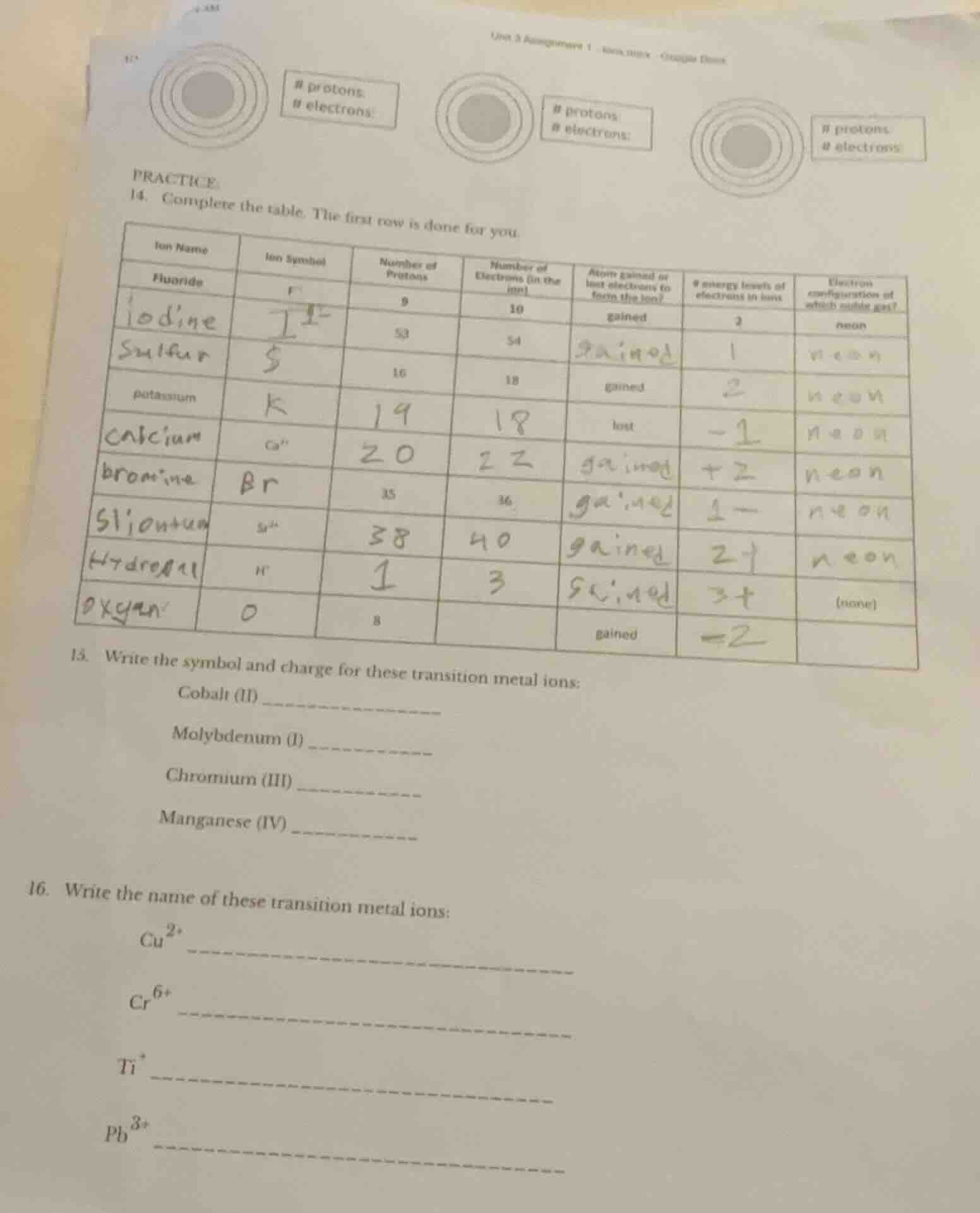
practice
- complete the table. the first row is done for you.
| ion name | ion symbol | number of protons | number of electrons (in the ion) | atom gained or lost electrons to form the ion? | # energy levels of electrons in ion | electron configuration of which noble gas? |
|---|---|---|---|---|---|---|
| iodine | 53 | 54 | 1 | |||
| sulfur | 16 | 18 | gained | |||
| potassium | lost | |||||
| calcium | ca²⁺ | 20 | ||||
| bromine | br⁻ | 35 | 36 | |||
| strontium | sr²⁺ | 38 | 40 | |||
| hydrogen | h⁺ | 1 | 0 | (none) | ||
| oxygen | o²⁻ | 8 | gained |
- write the symbol and charge for these transition metal ions:
cobalt (ii) ______
molybdenum (i) ______
chromium (iii) ______
manganese (iv) ______
- write the name of these transition metal ions:
cu²⁺ ______
cr⁶⁺ ______
ti⁺ ______
pb³⁺ ______
Question 15 (Writing symbol and charge for transition metal ions)
Step 1: Cobalt (II)
The Roman numeral (II) indicates the charge is +2. The symbol for cobalt is Co. So the ion is $\text{Co}^{2+}$.
Step 2: Molybdenum (I)
Roman numeral (I) means charge +1. Symbol for molybdenum is Mo. Ion: $\text{Mo}^{+}$.
Step 3: Chromium (III)
Roman numeral (III) gives charge +3. Symbol Cr. Ion: $\text{Cr}^{3+}$.
Step 4: Manganese (IV)
Roman numeral (IV) means charge +4. Symbol Mn. Ion: $\text{Mn}^{4+}$.
Question 16 (Writing name of transition metal ions)
Step 1: $\text{Cu}^{2+}$
Copper has a charge of +2, so name is Copper (II) ion.
Step 2: $\text{Cr}^{6+}$
Chromium with charge +6, name: Chromium (VI) ion.
Step 3: $\text{Ti}^{+}$
Titanium with charge +1, name: Titanium (I) ion.
Step 4: $\text{Pb}^{3+}$
Lead with charge +3, name: Lead (III) ion.
Question 14 (Completing the table - let's take Oxygen as an example)
For Oxygen (O):
- Number of protons = 8 (atomic number of O).
- Since it gained electrons, and for O, typical ion is $\text{O}^{2-}$, so number of electrons = 8 + 2 = 10.
- Energy levels: O has electron configuration related to neon? Wait, O gains 2 electrons to have 10 electrons, same as Ne (neon has 10 electrons). So electron configuration of which noble gas? Neon.
- Number of energy levels of electrons in ion: O$^{2-}$ has electron configuration 1s² 2s² 2p⁶, so 2 energy levels? Wait, original O has 8 electrons, 2 in first, 6 in second. After gaining 2, 8 in second? Wait no, 2 in first, 8 in second? Wait 2 + 8 = 10. So energy levels: 2.
So for Oxygen row:
- Ion Symbol: $\text{O}^{2-}$ (since it gained 2 electrons, charge -2)
- Number of Electrons: 10
- Electron configuration of which noble gas: Neon (Ne)
- # energy levels of electrons in ion: 2
Final Answers (for Question 15):
- Cobalt (II): $\boldsymbol{\text{Co}^{2+}}$
- Molybdenum (I): $\boldsymbol{\text{Mo}^{+}}$
- Chromium (III): $\boldsymbol{\text{Cr}^{3+}}$
- Manganese (IV): $\boldsymbol{\text{Mn}^{4+}}$
Final Answers (for Question 16):
- $\text{Cu}^{2+}$: Copper (II) ion
- $\text{Cr}^{6+}$: Chromium (VI) ion
- $\text{Ti}^{+}$: Titanium (I) ion
- $\text{Pb}^{3+}$: Lead (III) ion
Final Answers (for Question 14 - Oxygen row):
- Ion Symbol: $\text{O}^{2-}$
- Number of Electrons: 10
- Electron configuration of which noble gas: Neon
- # energy levels of electrons in ion: 2
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 15 (Writing symbol and charge for transition metal ions)
Step 1: Cobalt (II)
The Roman numeral (II) indicates the charge is +2. The symbol for cobalt is Co. So the ion is $\text{Co}^{2+}$.
Step 2: Molybdenum (I)
Roman numeral (I) means charge +1. Symbol for molybdenum is Mo. Ion: $\text{Mo}^{+}$.
Step 3: Chromium (III)
Roman numeral (III) gives charge +3. Symbol Cr. Ion: $\text{Cr}^{3+}$.
Step 4: Manganese (IV)
Roman numeral (IV) means charge +4. Symbol Mn. Ion: $\text{Mn}^{4+}$.
Question 16 (Writing name of transition metal ions)
Step 1: $\text{Cu}^{2+}$
Copper has a charge of +2, so name is Copper (II) ion.
Step 2: $\text{Cr}^{6+}$
Chromium with charge +6, name: Chromium (VI) ion.
Step 3: $\text{Ti}^{+}$
Titanium with charge +1, name: Titanium (I) ion.
Step 4: $\text{Pb}^{3+}$
Lead with charge +3, name: Lead (III) ion.
Question 14 (Completing the table - let's take Oxygen as an example)
For Oxygen (O):
- Number of protons = 8 (atomic number of O).
- Since it gained electrons, and for O, typical ion is $\text{O}^{2-}$, so number of electrons = 8 + 2 = 10.
- Energy levels: O has electron configuration related to neon? Wait, O gains 2 electrons to have 10 electrons, same as Ne (neon has 10 electrons). So electron configuration of which noble gas? Neon.
- Number of energy levels of electrons in ion: O$^{2-}$ has electron configuration 1s² 2s² 2p⁶, so 2 energy levels? Wait, original O has 8 electrons, 2 in first, 6 in second. After gaining 2, 8 in second? Wait no, 2 in first, 8 in second? Wait 2 + 8 = 10. So energy levels: 2.
So for Oxygen row:
- Ion Symbol: $\text{O}^{2-}$ (since it gained 2 electrons, charge -2)
- Number of Electrons: 10
- Electron configuration of which noble gas: Neon (Ne)
- # energy levels of electrons in ion: 2
Final Answers (for Question 15):
- Cobalt (II): $\boldsymbol{\text{Co}^{2+}}$
- Molybdenum (I): $\boldsymbol{\text{Mo}^{+}}$
- Chromium (III): $\boldsymbol{\text{Cr}^{3+}}$
- Manganese (IV): $\boldsymbol{\text{Mn}^{4+}}$
Final Answers (for Question 16):
- $\text{Cu}^{2+}$: Copper (II) ion
- $\text{Cr}^{6+}$: Chromium (VI) ion
- $\text{Ti}^{+}$: Titanium (I) ion
- $\text{Pb}^{3+}$: Lead (III) ion
Final Answers (for Question 14 - Oxygen row):
- Ion Symbol: $\text{O}^{2-}$
- Number of Electrons: 10
- Electron configuration of which noble gas: Neon
- # energy levels of electrons in ion: 2