QUESTION IMAGE
Question
practice unit 3 - spring 2026
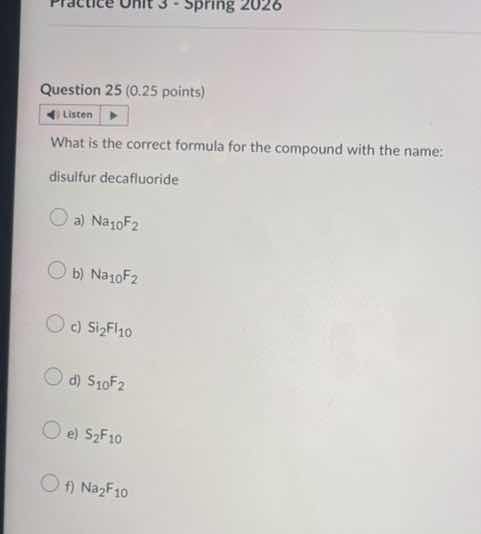
question 25 (0.25 points)
listen
what is the correct formula for the compound with the name:
disulfur decafluoride
a) $\ce{na_{10}f_{2}}$
b) $\ce{na_{10}f_{2}}$
c) $\ce{si_{2}f_{10}}$
d) $\ce{s_{10}f_{2}}$
e) $\ce{s_{2}f_{10}}$
f) $\ce{na_{2}f_{10}}$
Brief Explanations
- Use prefixes to find element counts:
- "di-" means 2, so there are 2 sulfur (S) atoms.
- "deca-" means 10, so there are 10 fluorine (F) atoms.
- Combine the elements with their counts in the order given by the name (sulfur first, then fluorine).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
e) $S_2F_{10}$