QUESTION IMAGE
Question
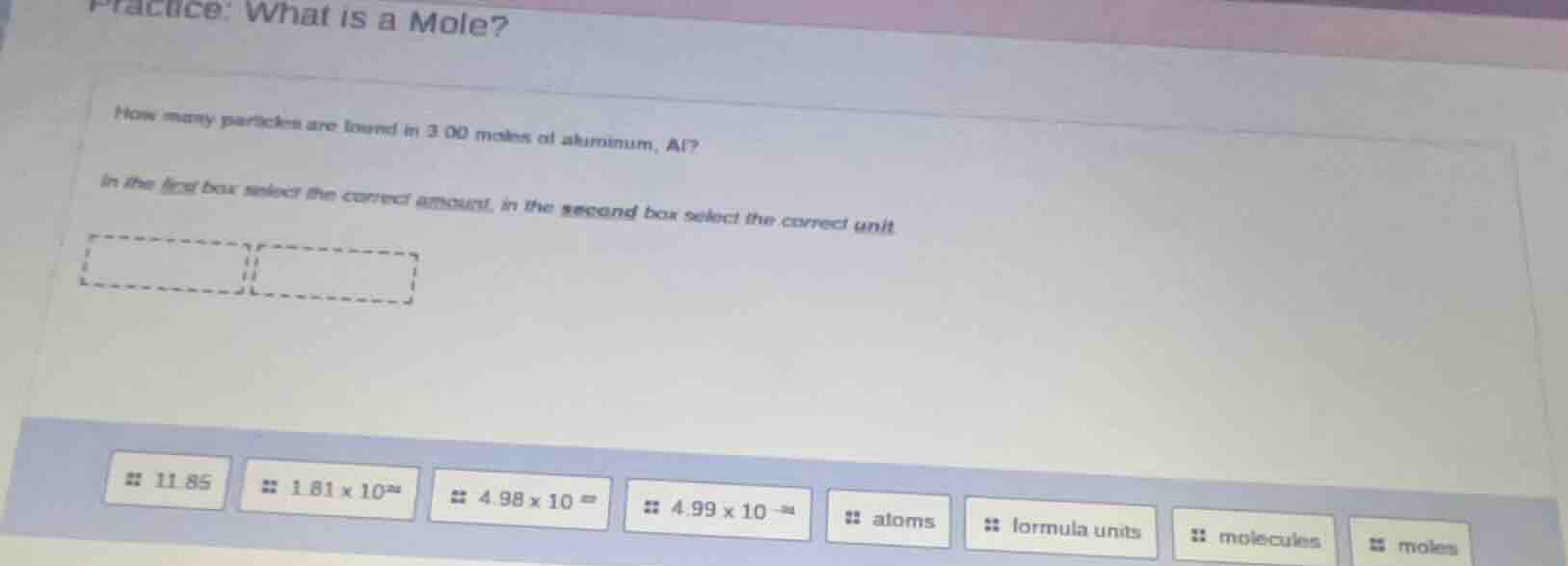
practice: what is a mole?
how many particles are found in 3.00 moles of aluminum, al?
in the first box select the correct amount, in the second box select the correct unit
11.85
1.81×10²⁴
4.98×10²³
4.99×10⁻²⁴
atoms
formula units
molecules
moles
Step1: Recall Avogadro's number
Avogadro's number is \( 6.022\times10^{23} \) particles per mole.
Step2: Calculate number of particles
To find the number of particles in 3.00 moles of aluminum, we use the formula: Number of particles = Moles × Avogadro's number.
So, Number of particles = \( 3.00\space mol\times6.022\times10^{23}\space \frac{particles}{mol} \)
\( = 1.8066\times10^{24}\approx1.81\times10^{24} \)
Since aluminum is an element, the particles are atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First box: \( 1.81\times10^{24} \)
Second box: atoms