QUESTION IMAGE
Question
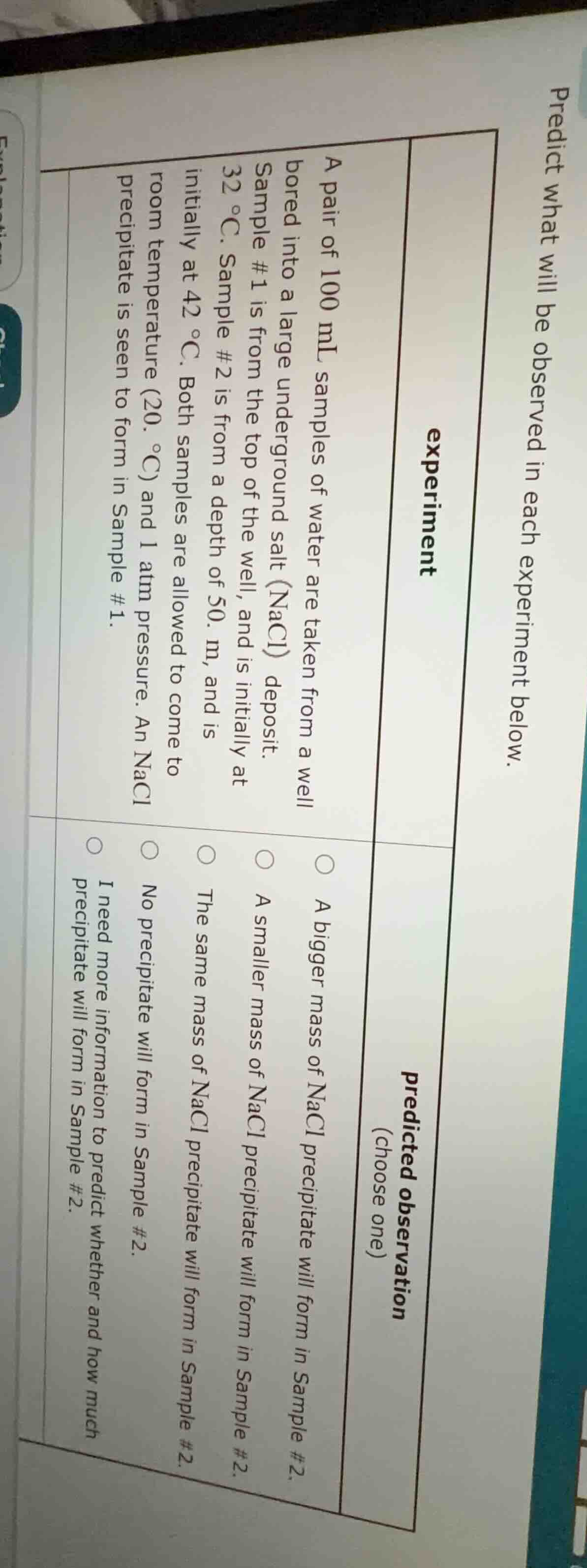
predict what will be observed in each experiment below.
experiment
a pair of 100 ml samples of water are taken from a well bored into a large underground salt (nacl) deposit. sample #1 is from the top of the well, and is initially at 32 °c. sample #2 is from a depth of 50. m, and is initially at 42 °c. both samples are allowed to come to room temperature (20. °c) and 1 atm pressure. an nacl precipitate is seen to form in sample #1.
predicted observation (choose one)
○ a bigger mass of nacl precipitate will form in sample #2.
○ a smaller mass of nacl precipitate will form in sample #2.
○ the same mass of nacl precipitate will form in sample #2.
○ no precipitate will form in sample #2.
○ i need more information to predict whether and how much precipitate will form in sample #2.
- First, note that the solubility of NaCl in water is nearly unaffected by temperature (only a very slight increase with higher temp) and pressure (negligible effect for condensed phases like aqueous solutions).
- Sample #1 was taken from a warm (32 °C) shallow well, and when cooled to 20 °C, NaCl precipitated, meaning it was saturated at 32 °C.
- Sample #2 was taken from a warmer (42 °C) deep well. Since NaCl solubility barely changes with temperature, the warm 42 °C water can hold almost the same amount of NaCl as the 32 °C water. When cooled to 20 °C, it will also become supersaturated to nearly the same degree as Sample #1, leading to the same mass of precipitate forming.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The same mass of NaCl precipitate will form in Sample #2.