QUESTION IMAGE
Question
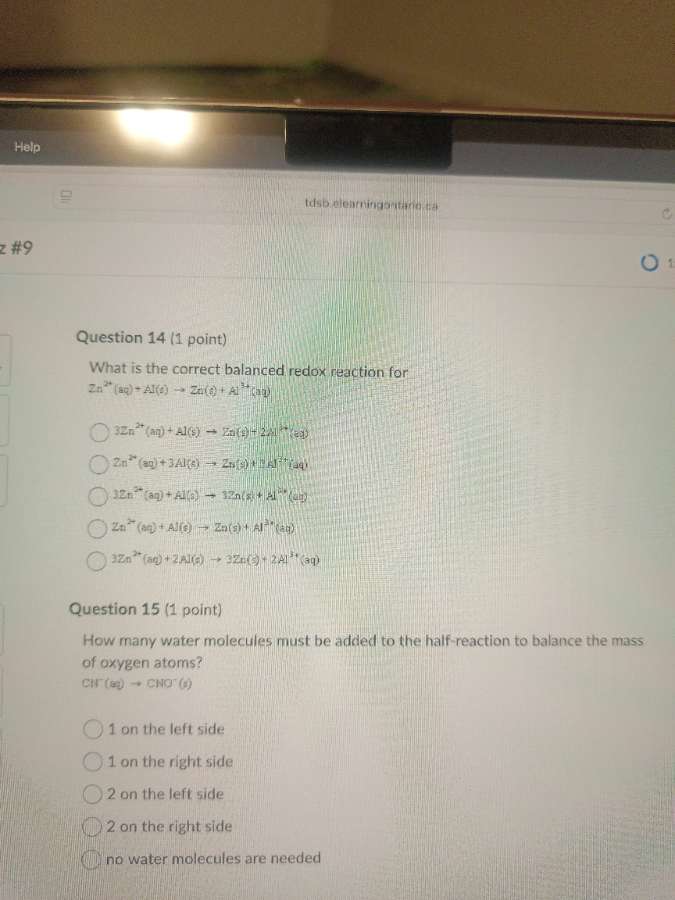
question 14 (1 point)
what is the correct balanced redox reaction for
zn²⁺(aq) + al(s) → zn(s) + al³⁺(aq)
options:
3zn²⁺(aq) + al(s) → zn(s) + 2al³⁺(aq)
zn²⁺(aq) + 3al(s) → zn(s) + 3al³⁺(aq)
3zn²⁺(aq) + al(s) → 3zn(s) + al³⁺(aq)
zn²⁺(aq) + al(s) → zn(s) + al³⁺(aq)
3zn²⁺(aq) + 2al(s) → 3zn(s) + 2al³⁺(aq)
question 15 (1 point)
how many water molecules must be added to the half - reaction to balance the mass of oxygen atoms?
cn⁻(aq) → cno⁻(g)
options:
1 on the left side
1 on the right side
2 on the left side
2 on the right side
no water molecules are needed
Question 14
Step1: Determine oxidation states
- Zn in \( Zn^{2+} \) has +2, becomes Zn(0) (reduction, gain 2 e⁻).
- Al(0) becomes \( Al^{3+} \) (oxidation, lose 3 e⁻).
Step2: Balance electrons
- LCM of 2 and 3 is 6. So, 3 \( Zn^{2+} \) (gain \( 3\times2 = 6 \) e⁻) and 2 Al (lose \( 2\times3 = 6 \) e⁻).
Step3: Write balanced reaction
- Multiply \( Zn^{2+} \) by 3, Al by 2. Products: 3 Zn(s) and 2 \( Al^{3+} \)(aq).
- Reaction: \( 3Zn^{2+}(aq) + 2Al(s)
ightarrow 3Zn(s) + 2Al^{3+}(aq) \)
Step1: Analyze oxygen in reactant and product
- Reactant: \( CN^-(aq) \) has 0 O. Product: \( CNO^-(aq) \) has 1 O.
Step2: Balance O with \( H_2O \)
- To add 1 O to left (reactant side) or right (product side)? Wait, \( CN^-
ightarrow CNO^- \): need 1 O on left? No, \( H_2O \) has O. Wait, \( CN^- \) (no O) → \( CNO^- \) (1 O). So add 1 \( H_2O \) to left (reactant side) to provide 1 O. Wait, no: \( CN^- + H_2O
ightarrow CNO^- +... \) (O from \( H_2O \)). Wait, let's check:
- Reactant: \( CN^- \) (0 O), \( H_2O \) (1 O) → total O:1. Product: \( CNO^- \) (1 O). So add 1 \( H_2O \) on left. Wait, no: the half - reaction is \( CN^-(aq)
ightarrow CNO^-(aq) \). Wait, mass balance for O: product has 1 O, reactant has 0. So add 1 \( H_2O \) to left (reactant side) to get 1 O.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 3Zn^{2+}(aq) + 2Al(s)
ightarrow 3Zn(s) + 2Al^{3+}(aq) \) (the last option)