QUESTION IMAGE
Question
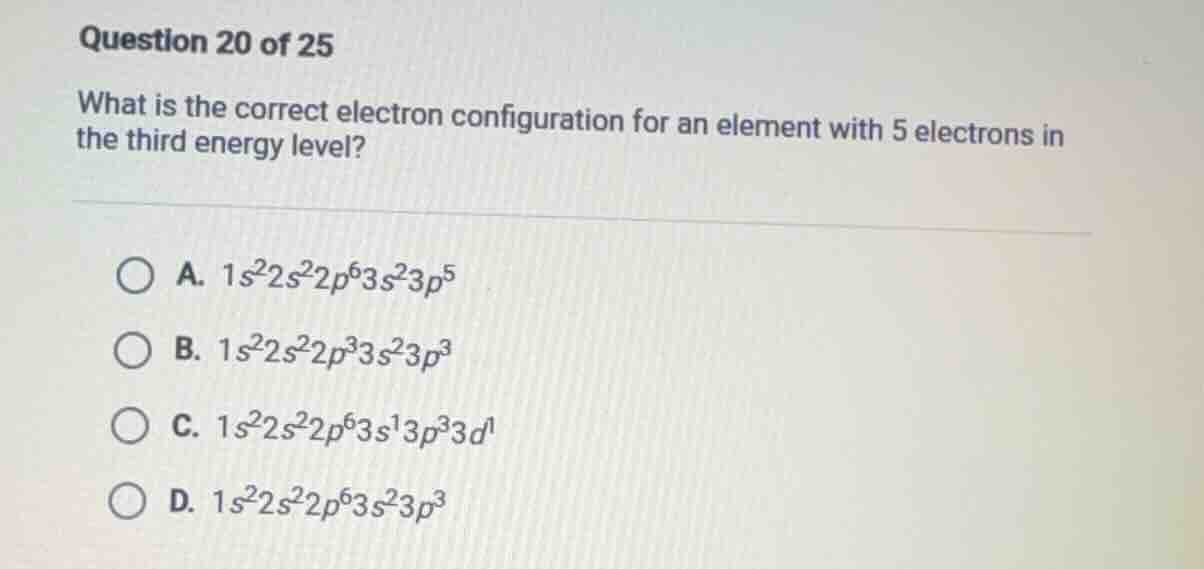
question 20 of 25
what is the correct electron configuration for an element with 5 electrons in the third energy level?
a. $1s^2 2s^2 2p^6 3s^2 3p^5$
b. $1s^2 2s^2 2p^3 3s^2 3p^3$
c. $1s^2 2s^2 2p^6 3s^1 3p^3 3d^1$
d. $1s^2 2s^2 2p^6 3s^2 3p^3$
Brief Explanations
- Recall electron configuration rules: Fill orbitals in order \(1s \to 2s \to 2p \to 3s \to 3p \to 4s \to 3d \dots\). The \(s\)-orbital holds 2, \(p\)-orbital holds 6, \(d\)-orbital holds 10 electrons.
- Analyze the third energy level (\(n = 3\)): It includes \(3s\), \(3p\), and \(3d\) (but \(3d\) fills after \(4s\), so for 5 electrons in \(n = 3\), first fill \(3s\) (2 electrons), then \(3p\) (remaining \(5 - 2=3\) electrons).
- Check each option:
- Option A: \(3p^5\) means 2 (from \(3s\)) + 5 (from \(3p\)) = 7 electrons in \(n = 3\) → Incorrect.
- Option B: \(2p^3\) is wrong ( \(2p\) should have 6 electrons to fill before \(3s\)) → Incorrect.
- Option C: \(3s^1\) and \(3d^1\) are wrong ( \(3s\) should have 2, \(3d\) fills after \(4s\)) → Incorrect.
- Option D: \(3s^2\) (2 electrons) + \(3p^3\) (3 electrons) = 5 electrons in \(n = 3\), and \(1s^22s^22p^6\) is correct filling for \(n = 1,2\) → Correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. \(1s^22s^22p^63s^23p^3\)