QUESTION IMAGE
Question
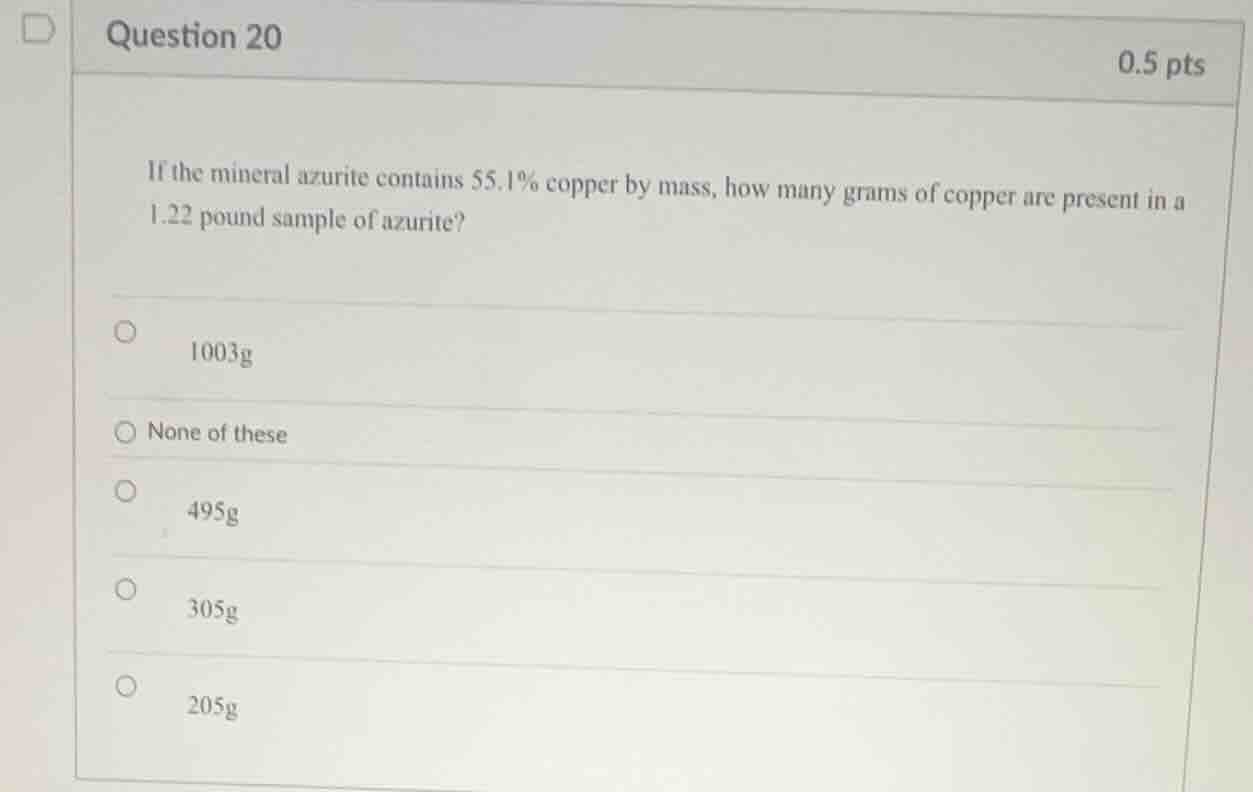
question 20 0.5 pts if the mineral azurite contains 55.1% copper by mass, how many grams of copper are present in a 1.22 pound sample of azurite? 1003g none of these 495g 305g 205g
Step1: Convert pounds to grams
We know that 1 pound = 453.592 grams. So for 1.22 pounds, the mass in grams is $1.22\times453.592$ grams.
$1.22\times453.592 = 553.38224$ grams (mass of azurite sample in grams)
Step2: Calculate mass of copper
The mass of copper is 55.1% of the mass of azurite. So we calculate $55.1\%$ of $553.38224$ grams.
First, convert 55.1% to decimal: $55.1\%=0.551$
Then, mass of copper = $0.551\times553.38224$
$0.551\times553.38224\approx305$ grams
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
305g (corresponding to the option with 305g)