QUESTION IMAGE
Question
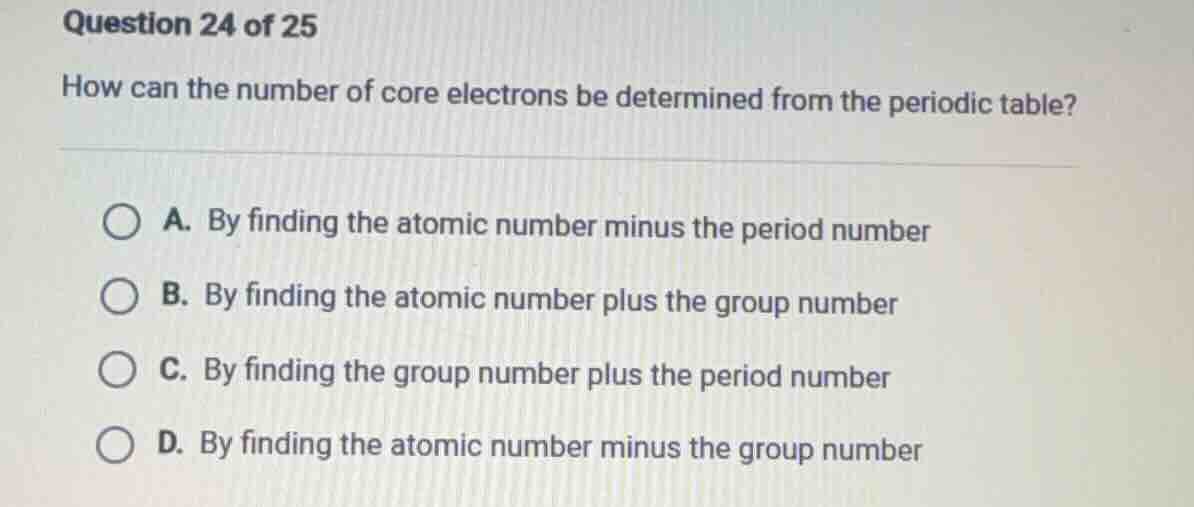
question 24 of 25
how can the number of core electrons be determined from the periodic table?
a. by finding the atomic number minus the period number
b. by finding the atomic number plus the group number
c. by finding the group number plus the period number
d. by finding the atomic number minus the group number
Core electrons are the electrons in an atom that are not valence electrons. Valence electrons are related to the group number (for main - group elements, the number of valence electrons is equal to the group number, or for groups 13 - 18, it is group number - 10). The atomic number (Z) is the total number of electrons in a neutral atom. So, to find the number of core electrons, we can use the formula: number of core electrons = atomic number - number of valence electrons. Since the number of valence electrons is related to the group number (for main - group elements), the number of core electrons can be determined by atomic number minus group number. Let's analyze each option:
- Option A: Atomic number minus period number has no relation to the calculation of core electrons. The period number is related to the number of electron shells, not core electrons.
- Option B: Atomic number plus group number will give a value that is not related to core electrons.
- Option C: Group number plus period number is also not related to the number of core electrons.
- Option D: As explained above, atomic number minus group number (for main - group elements, where group number gives the number of valence electrons) gives the number of core electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. By finding the atomic number minus the group number