QUESTION IMAGE
Question
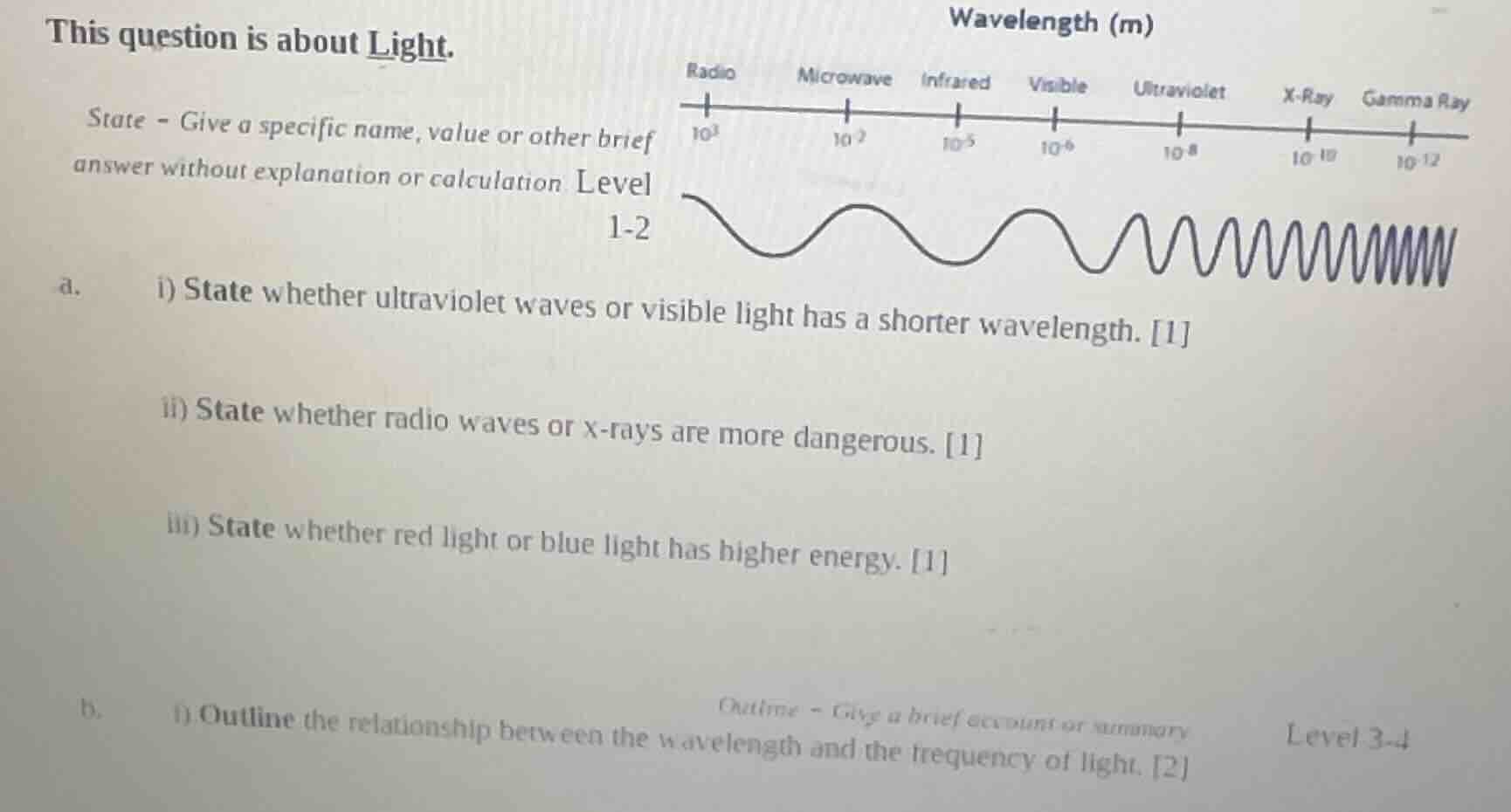
this question is about light.
state - give a specific name, value or other brief answer without explanation or calculation level 1-2
a. i) state whether ultraviolet waves or visible light has a shorter wavelength. 1
ii) state whether radio waves or x-rays are more dangerous. 1
iii) state whether red light or blue light has higher energy. 1
b. i) outline the relationship between the wavelength and the frequency of light. 2
outline - give a brief account or summary level 3-4
Part a
i)
From the electromagnetic spectrum (wavelength scale), ultraviolet has a wavelength around \(10^{-8}\) m and visible light around \(10^{-6}\) m. Smaller value means shorter wavelength. So ultraviolet waves have shorter wavelength.
X - rays have higher energy (shorter wavelength, higher frequency) than radio waves. Higher - energy electromagnetic waves are more dangerous as they can ionize atoms/molecules. So X - rays are more dangerous.
Blue light has a shorter wavelength than red light. Using the formula \(E = hf=\frac{hc}{\lambda}\) (where \(E\) is energy, \(h\) is Planck's constant, \(f\) is frequency, \(c\) is speed of light, \(\lambda\) is wavelength), shorter wavelength means higher energy. So blue light has higher energy.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Ultraviolet waves