QUESTION IMAGE
Question
question 5
mark this question
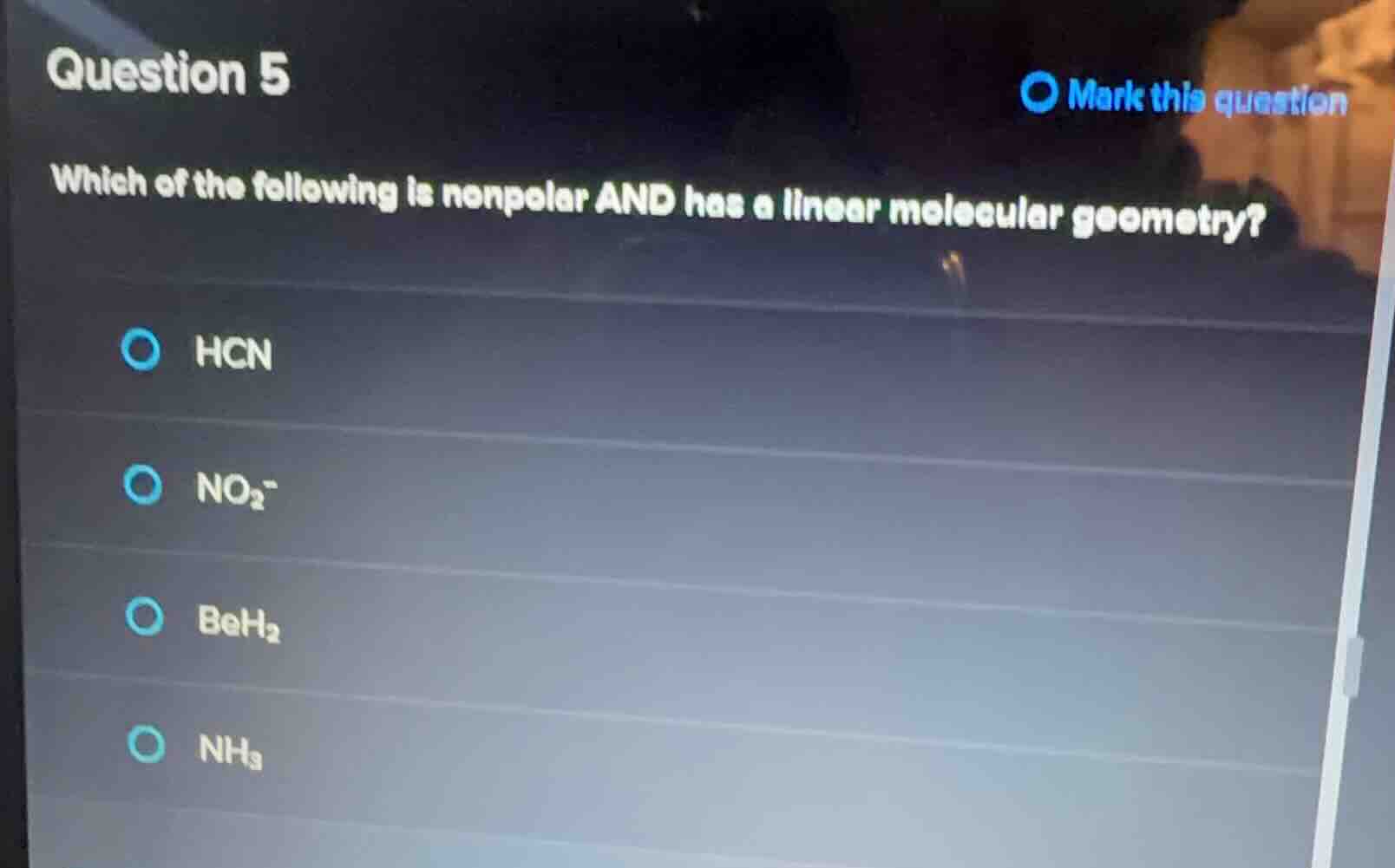
which of the following is nonpolar and has a linear molecular geometry?
hcn
no₂⁻
beh₂
nh₃
Brief Explanations
- HCN: Has linear geometry, but is polar because C-H and C-N bonds have unequal electronegativity differences, creating a net dipole.
- NO₂⁻: Has bent (V-shaped) molecular geometry due to a lone pair on N, so it is not linear, and is polar.
- BeH₂: Has linear geometry (central Be has no lone pairs, bonded to two H atoms). The symmetric linear arrangement cancels out the small bond dipoles, making it nonpolar.
- NH₃: Has trigonal pyramidal geometry, so it is not linear, and is polar due to the lone pair on N.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
BeH₂