QUESTION IMAGE
Question
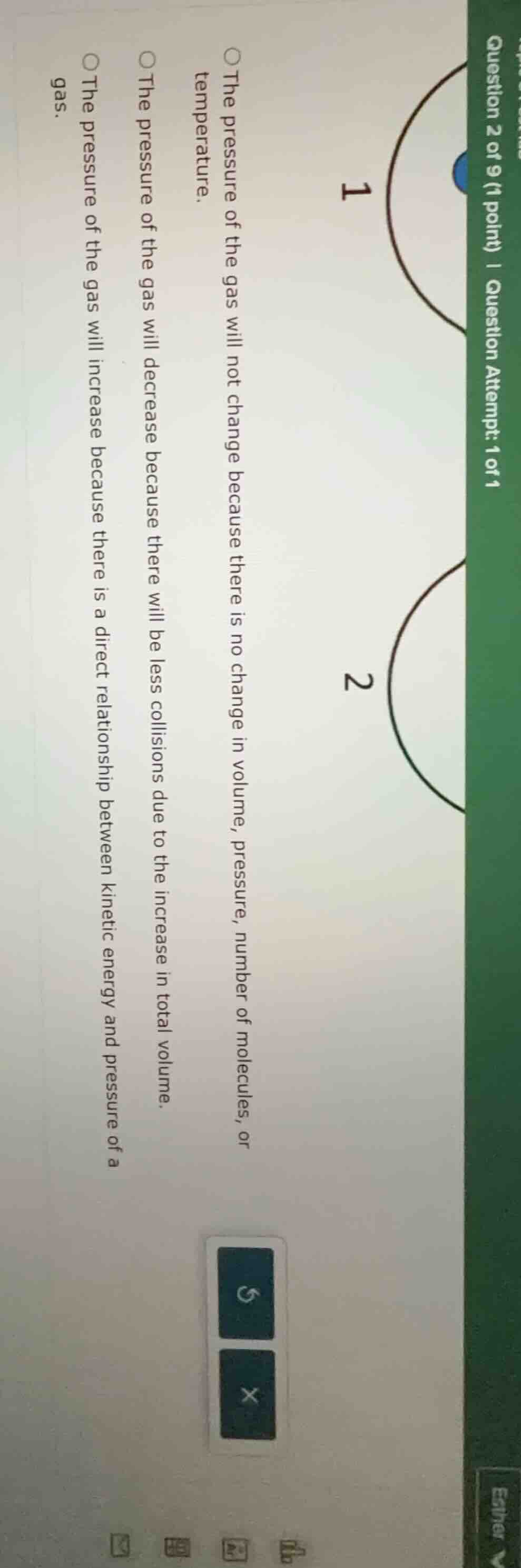
question 2 of 9 (1 point) | question attempt: 1 of 1 1 2 the pressure of the gas will not change because there is no change in volume, pressure, number of molecules, or temperature. the pressure of the gas will decrease because there will be less collisions due to the increase in total volume. the pressure of the gas will increase because there is a direct relationship between kinetic energy and pressure of a gas.
To solve this, we analyze gas laws (like Boyle's, Charles's, or kinetic theory). The second option says pressure decreases with increased volume (more space, fewer collisions) – this matches Boyle’s law (P ∝ 1/V, constant temp, moles). The first is wrong (no temp change info), third is wrong (volume change here, and pressure - volume are related). So the correct reasoning is about volume - pressure inverse relation (more volume → fewer collisions → lower pressure).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The pressure of the gas will decrease because there will be less collisions due to the increase in total volume.