QUESTION IMAGE
Question
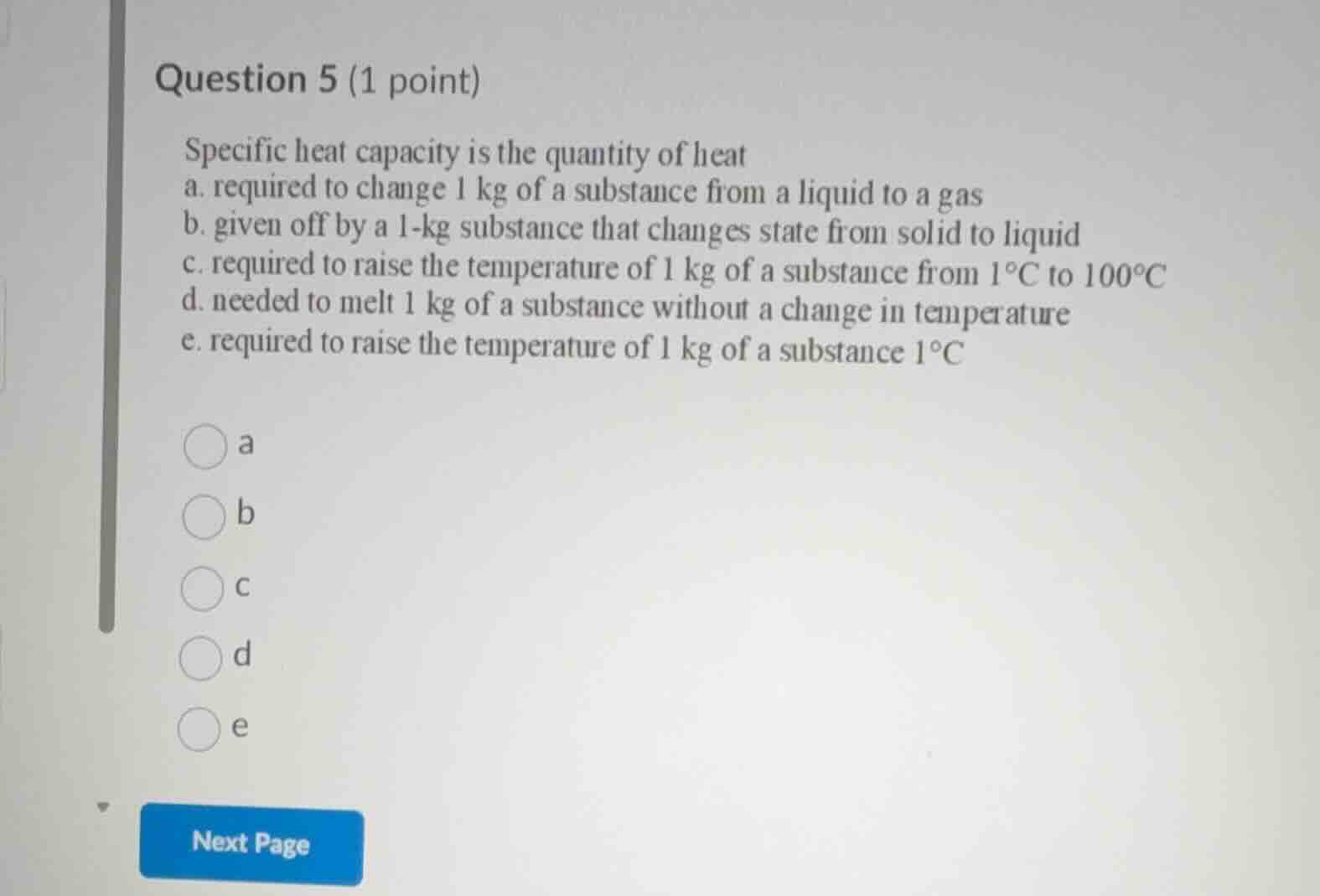
question 5 (1 point)
specific heat capacity is the quantity of heat
a. required to change 1 kg of a substance from a liquid to a gas
b. given off by a 1-kg substance that changes state from solid to liquid
c. required to raise the temperature of 1 kg of a substance from 1°c to 100°c
d. needed to melt 1 kg of a substance without a change in temperature
e. required to raise the temperature of 1 kg of a substance 1°c
a
b
c
d
e
Brief Explanations
- Option a: Describes latent heat of vaporization (liquid to gas), not specific heat capacity.
- Option b: Describes latent heat of fusion (solid to liquid, releasing heat is incorrect here as fusion absorbs heat, also not specific heat).
- Option c: Specific heat is per 1°C temperature change, not from 1°C to 100°C (this is a range, not the definition).
- Option d: Describes latent heat of fusion (melting without temp change), not specific heat.
- Option e: Matches the definition of specific heat capacity: the heat required to raise the temperature of 1 kg of a substance by 1°C.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
e. required to raise the temperature of 1 kg of a substance 1°C