QUESTION IMAGE
Question
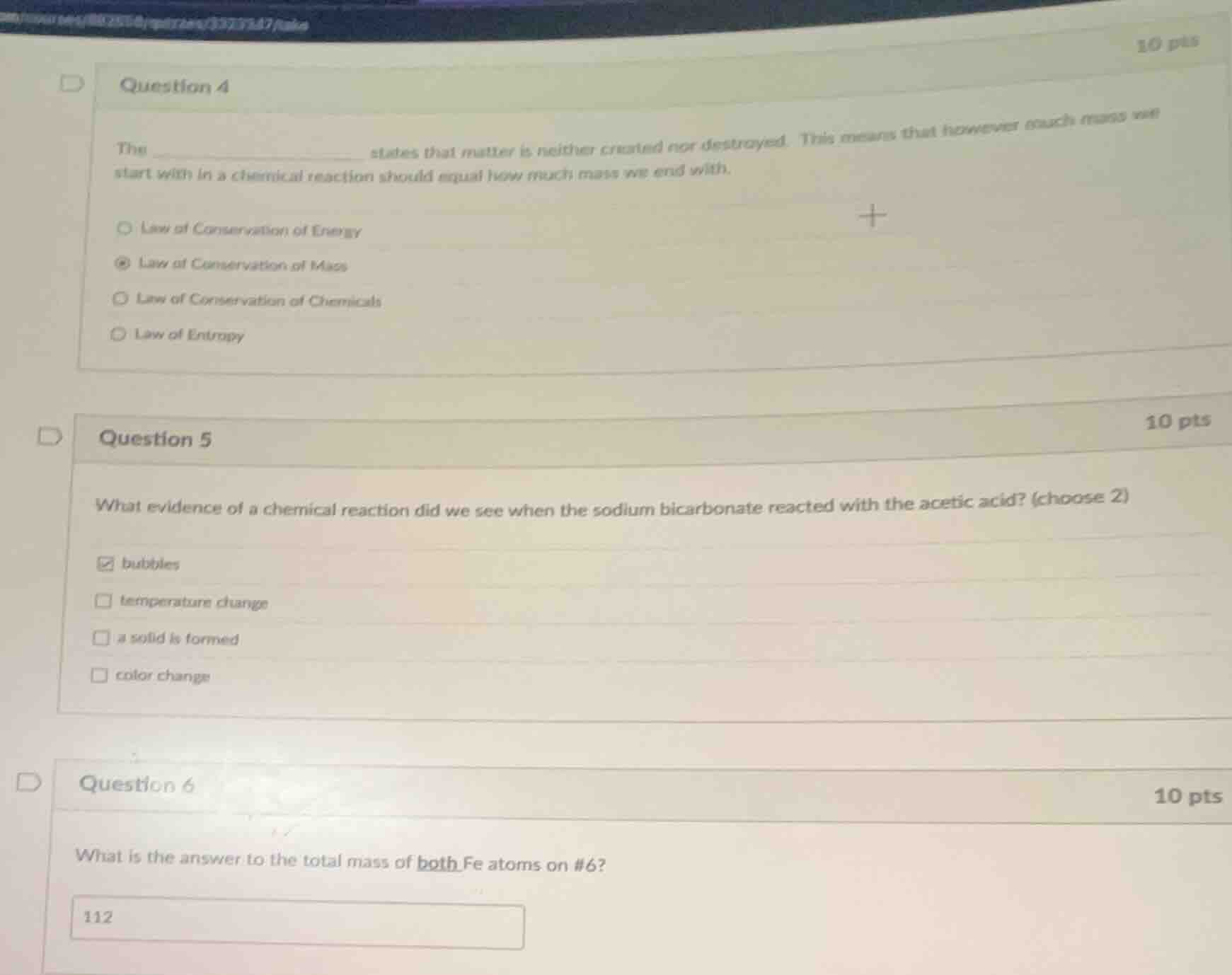
question 4
the ______________ states that matter is neither created nor destroyed. this means that however much mass we start with in a chemical reaction should equal how much mass we end with.
- law of conservation of energy
- law of conservation of mass
- law of conservation of chemicals
- law of entropy
question 5
what evidence of a chemical reaction did we see when the sodium bicarbonate reacted with the acetic acid? (choose 2)
- bubbles
- temperature change
- a solid is formed
- color change
question 6
what is the answer to the total mass of both fe atoms on #6?
112
Question 4:
The described law defines that mass is conserved in chemical reactions, matching the Law of Conservation of Mass.
Question 5:
When sodium bicarbonate reacts with acetic acid, carbon dioxide gas (bubbles) is produced, and the reaction is exothermic, causing a temperature change.
Question 6:
The molar mass of Fe is 56 g/mol. For 2 Fe atoms, total mass is $2 \times 56 = 112$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Question 4: Law of Conservation of Mass
- Question 5: bubbles, temperature change
- Question 6: 112