QUESTION IMAGE
Question
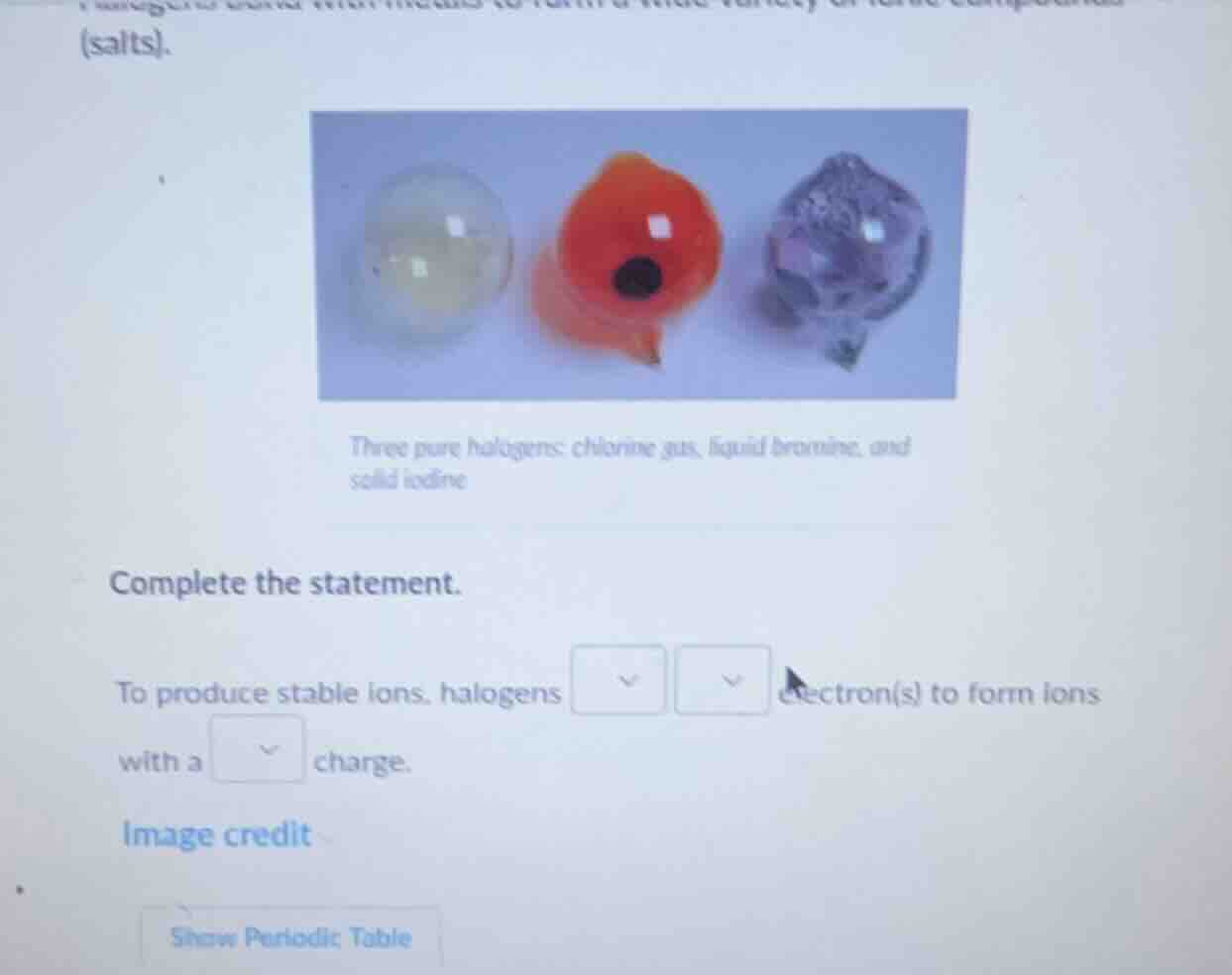
(salts).
three pure halogens: chlorine gas, liquid bromine, and solid iodine
complete the statement.
to produce stable ions, halogens electron(s) to form ions with a charge.
Halogens are in Group 17 of the periodic table, having 7 valence electrons. To achieve a stable octet (8 valence electrons), they gain 1 electron. Gaining an electron gives the ion a negative charge (since electrons are negative, adding one makes the ion have more negative charge than positive from protons). So the first dropdown is "gain", the second is "1", and the third is "negative".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First dropdown: gain
Second dropdown: 1
Third dropdown: negative