QUESTION IMAGE
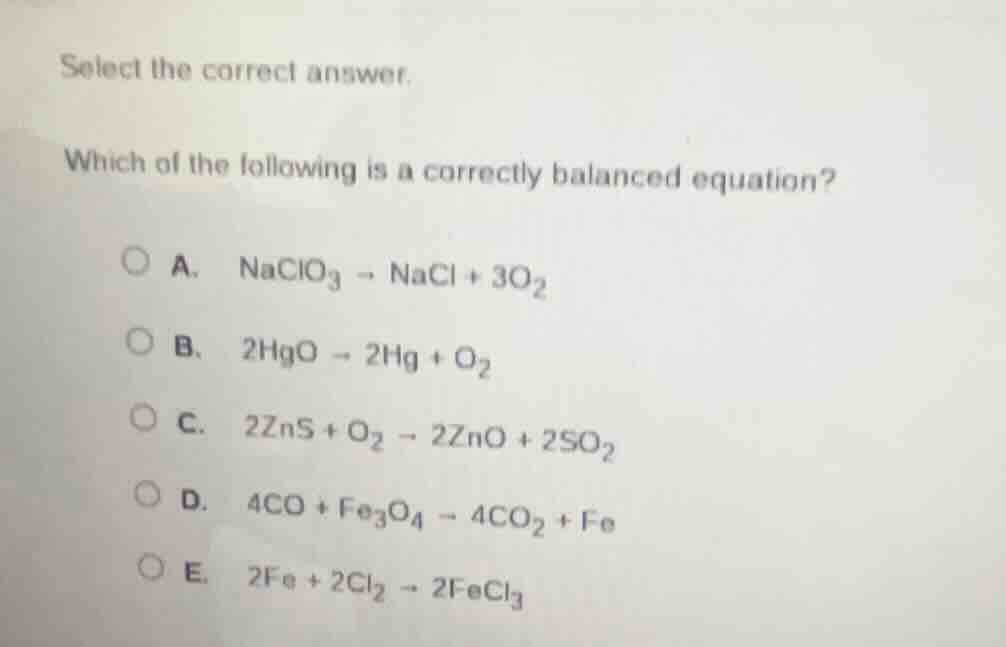
Question
select the correct answer.
which of the following is a correctly balanced equation?
a. $naclo_3 \
ightarrow nacl + 3o_2$
b. $2hgo \
ightarrow 2hg + o_2$
c. $2zns + o_2 \
ightarrow 2zno + 2so_2$
d. $4co + fe_3o_4 \
ightarrow 4co_2 + fe$
e. $2fe + 2cl_2 \
ightarrow 2fecl_3$
Step1: Check O balance for Option A
Reactant: $3$ O in $\text{NaClO}_3$; Product: $6$ O in $3\text{O}_2$. Unbalanced.
Step2: Check all atoms for Option B
Reactant: $2$ Hg, $2$ O in $2\text{HgO}$; Product: $2$ Hg, $2$ O in $2\text{Hg} + \text{O}_2$. Balanced.
Step3: Check S/O balance for Option C
Reactant: $2$ S, $2$ O in $2\text{ZnS} + \text{O}_2$; Product: $2$ S, $6$ O in $2\text{ZnO} + 2\text{SO}_2$. Unbalanced.
Step4: Check Fe balance for Option D
Reactant: $3$ Fe in $\text{Fe}_3\text{O}_4$; Product: $1$ Fe. Unbalanced.
Step5: Check Cl balance for Option E
Reactant: $4$ Cl in $2\text{Cl}_2$; Product: $6$ Cl in $2\text{FeCl}_3$. Unbalanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $2\text{HgO}
ightarrow 2\text{Hg} + \text{O}_2$