QUESTION IMAGE
Question
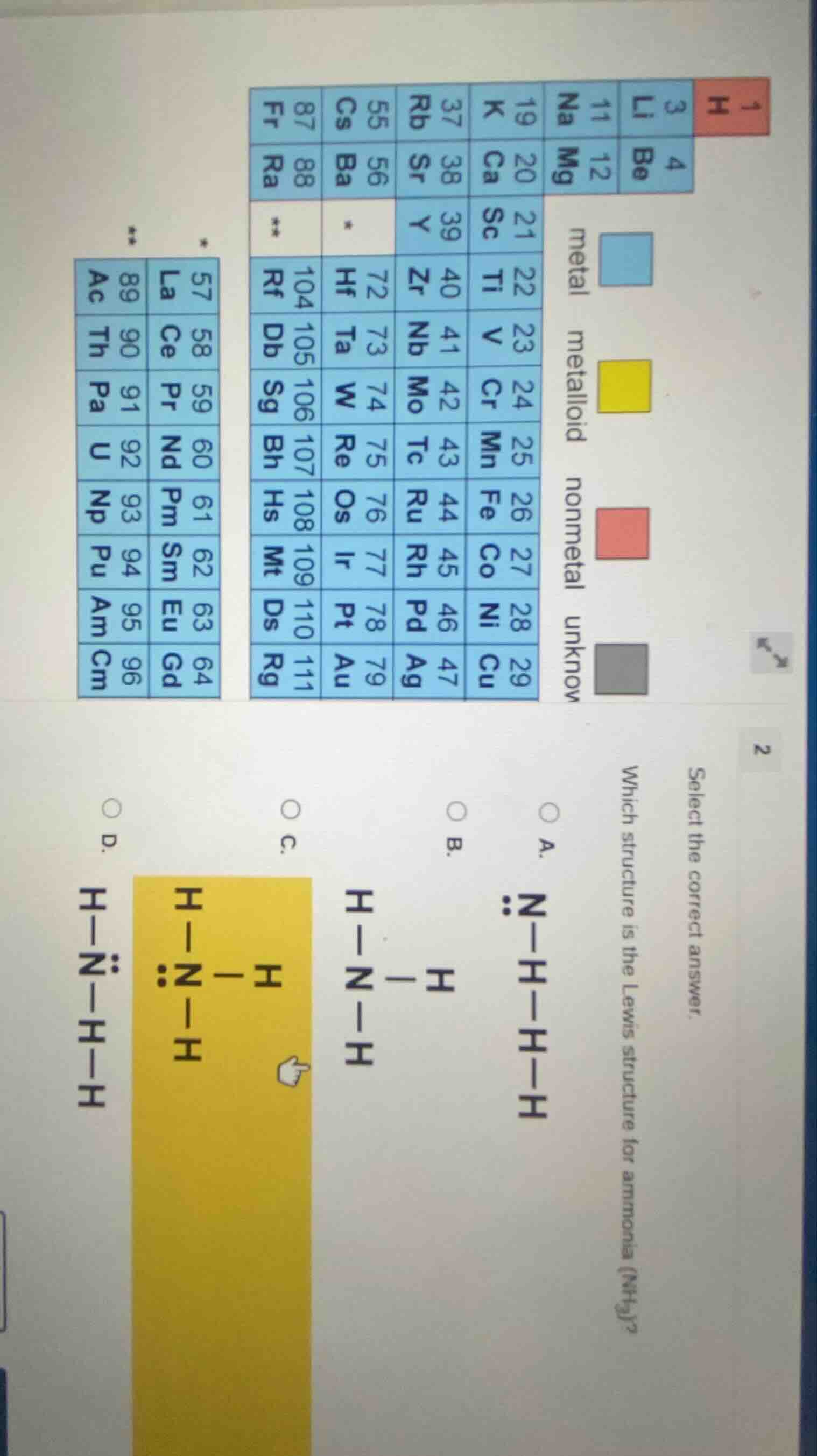
select the correct answer. which structure is the lewis structure for ammonia (nh₃)? a.
vdots n - h - h - h
b.
h - n - h
|
h
c.
h - n - h
|
h
vdots
d.
h - n - h - h
To determine the correct Lewis structure for ammonia ($\ce{NH_3}$), we analyze the valence electrons:
- Nitrogen (N) has 5 valence electrons.
- Each Hydrogen (H) has 1 valence electron, and there are 3 H atoms, so $3\times1 = 3$ valence electrons from H.
- Total valence electrons: $5 + 3 = 8$.
In a Lewis structure, we want to satisfy the octet rule for N (8 electrons) and duet rule for H (2 electrons each).
- Nitrogen forms single bonds with 3 H atoms, using $3\times2 = 6$ electrons (3 bonds).
- The remaining electrons on N: $8 - 6 = 2$, which form a lone pair (2 electrons).
Now let's analyze the options:
- Option A: The N has a double bond? No, $\ce{NH_3}$ has single bonds. Also, the lone pair placement is incorrect.
- Option B: No lone pair on N? Incorrect, N should have a lone pair.
- Option C: N is bonded to 3 H atoms (single bonds) and has a lone pair (2 electrons). This satisfies the valence electron count and octet/duet rules.
- Option D: Incorrect bonding (extra bond or incorrect lone pair).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $\ce{H - \overset{..}{N} - H}$ (with a third H bonded to N, as shown in the option's structure with N having a lone pair and three single bonds to H)