QUESTION IMAGE
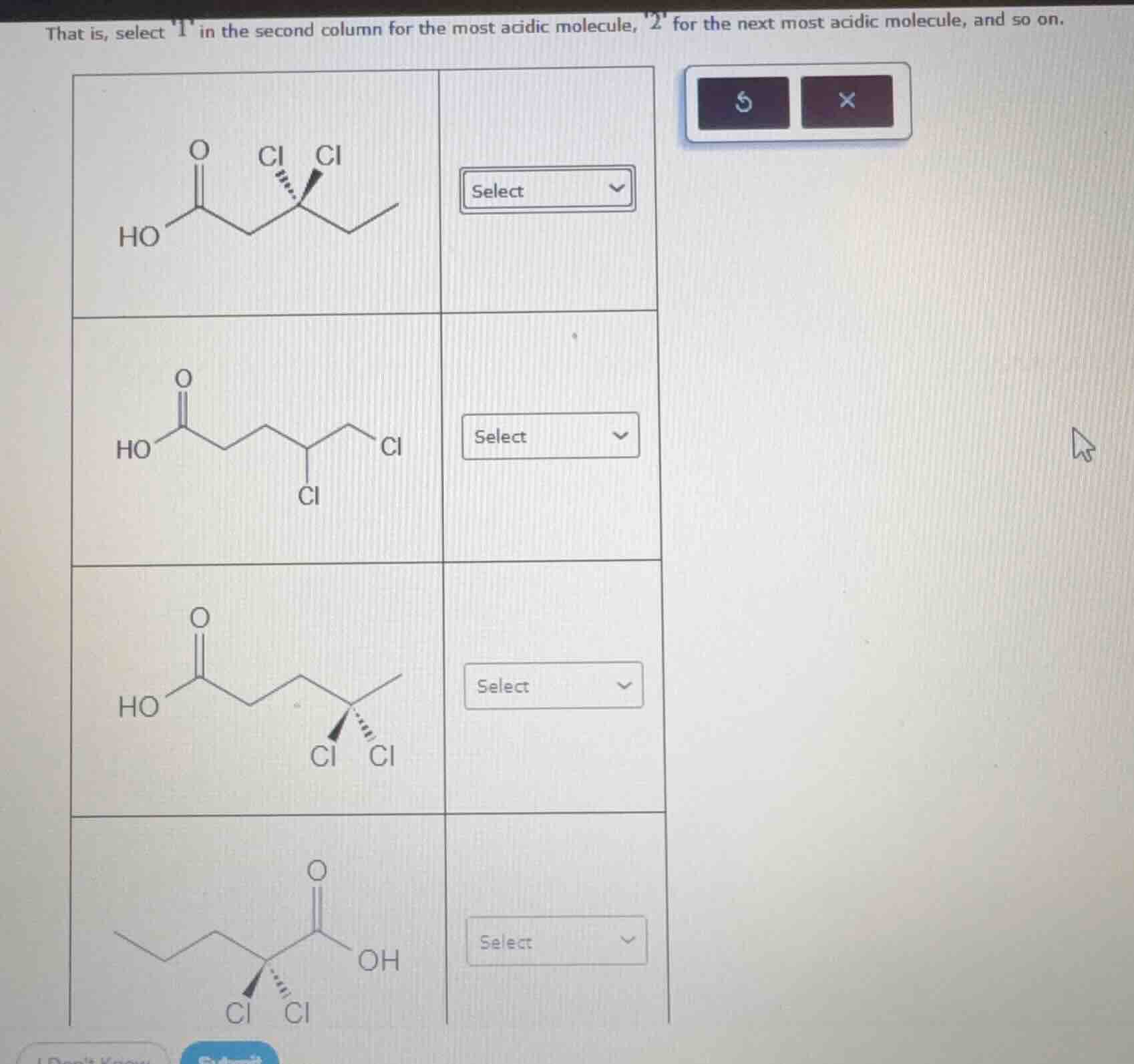
Question
that is, select 1 in the second column for the most acidic molecule, 2 for the next most acidic molecule, and so on.
To determine the acidity of these carboxylic acid molecules, we use the inductive effect (electron-withdrawing groups like $\ce{Cl}$ stabilize the conjugate base by dispersing negative charge). The key factors are:
- Proximity of $\boldsymbol{\ce{Cl}}$ to the carboxyl group (closer $\ce{Cl}$ atoms have a stronger inductive effect).
- Number of $\boldsymbol{\ce{Cl}}$ atoms (more $\ce{Cl}$ atoms increase electron withdrawal).
Step 1: Analyze Each Molecule
- Top molecule (first row): Two $\ce{Cl}$ atoms on the carbon adjacent to the $\ce{CH2}$ group (one carbon away from the carboxyl group, $\ce{-COOH}$).
- Second molecule (second row): Two $\ce{Cl}$ atoms, but one $\ce{Cl}$ is two carbons away from $\ce{-COOH}$, and the other is three carbons away.
- Third molecule (third row): Two $\ce{Cl}$ atoms on the carbon adjacent to the $\ce{CH2}$ group (one carbon away from $\ce{-COOH}$).
- Bottom molecule (fourth row): Two $\ce{Cl}$ atoms on the carbon directly attached to the carboxyl group (the $\alpha$-carbon, adjacent to $\ce{-COOH}$).
Step 2: Rank by Acidity
Acidity increases with:
- Closer $\ce{Cl}$ to $\ce{-COOH}$ (stronger inductive effect).
- More $\ce{Cl}$ atoms (greater electron withdrawal).
The bottom molecule has $\ce{Cl}$ atoms on the $\alpha$-carbon (closest to $\ce{-COOH}$), so it is the most acidic (rank 1).
The third and top molecules have $\ce{Cl}$ atoms one carbon away from $\ce{-COOH}$, but the third molecule’s $\ce{Cl}$ atoms are on a more substituted carbon (but inductive effect depends on distance, not substitution). However, the bottom molecule is still closest.
The second molecule has $\ce{Cl}$ atoms farther away, so it is the least acidic (rank 4).
Final Ranking (Most to Least Acidic)
- Bottom molecule (fourth row: $\ce{Cl}$ on $\alpha$-carbon).
- Third molecule (third row: $\ce{Cl}$ one carbon from $\ce{-COOH}$).
- Top molecule (first row: $\ce{Cl}$ one carbon from $\ce{-COOH}$).
- Second molecule (second row: $\ce{Cl}$ farther from $\ce{-COOH}$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Example Selection)
- Bottom molecule: Select $\boldsymbol{1}$ (most acidic).
- Third molecule: Select $\boldsymbol{2}$.
- Top molecule: Select $\boldsymbol{3}$.
- Second molecule: Select $\boldsymbol{4}$.
(Adjust based on exact molecular structure, but the key is proximity of $\ce{Cl}$ to $\ce{-COOH}$.)