QUESTION IMAGE
Question
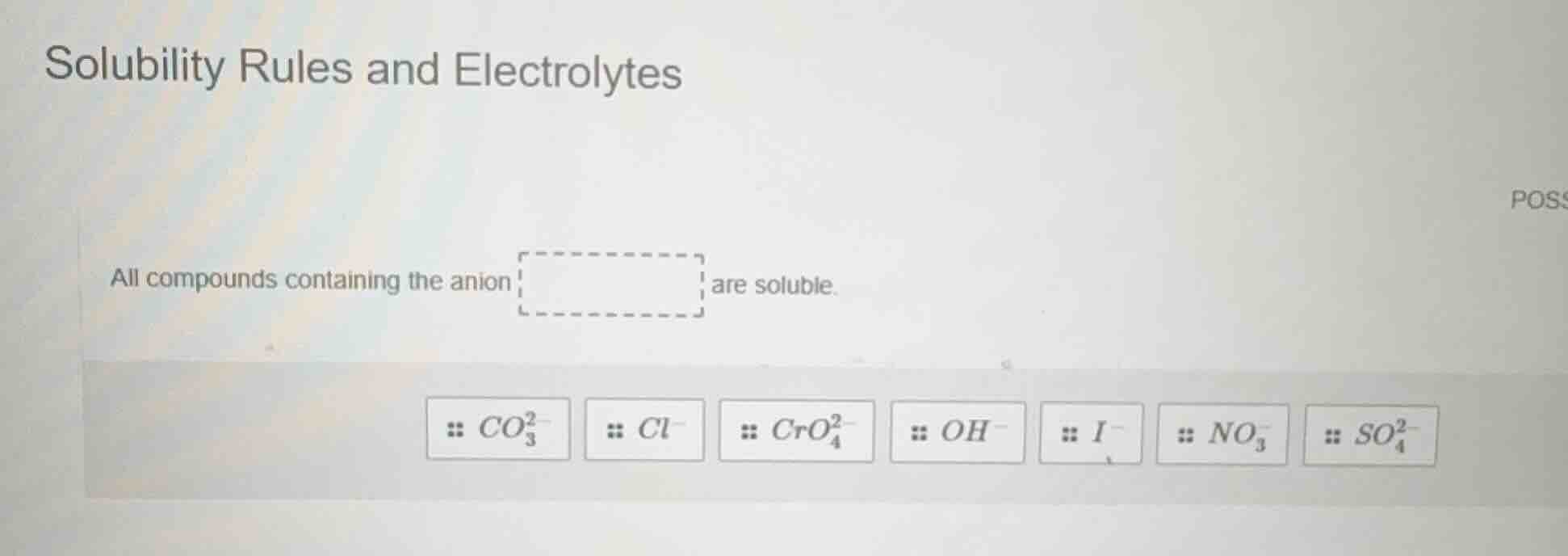
solubility rules and electrolytes
all compounds containing the anion are soluble.
$co_{3}^{2-}$ $cl^{-}$ $cro_{4}^{2-}$ $oh^{-}$ $i^{-}$ $no_{3}^{-}$ $so_{4}^{2-}$
According to standard solubility rules for ionic compounds, nitrate salts (containing the $\boldsymbol{NO_3^-}$ anion) are universally soluble in water, with no common exceptions. Other anions listed have solubility exceptions: chloride/iodide have insoluble salts with $Ag^+, Pb^{2+}, Hg_2^{2+}$; carbonate, chromate, hydroxide have most insoluble salts with non-group 1/ammonium cations; sulfate has insoluble salts with $Ba^{2+}, Pb^{2+}, Ca^{2+}$ etc.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{NO_3^-}$