QUESTION IMAGE
Question
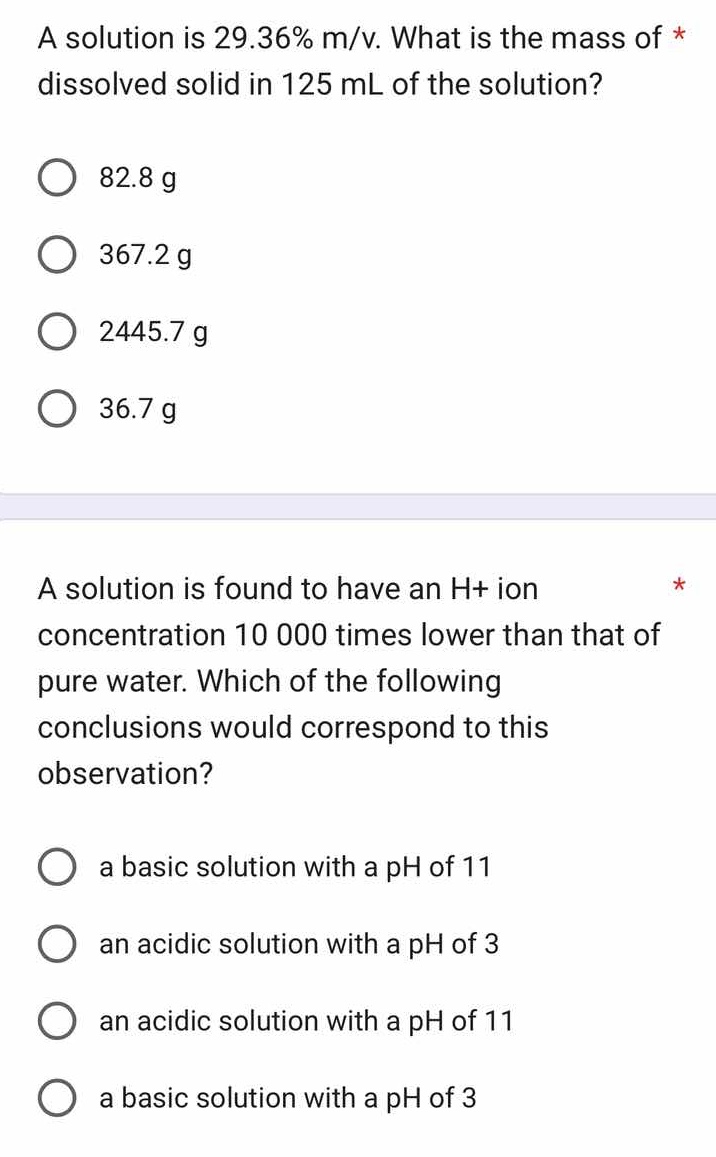
a solution is 29.36% m/v. what is the mass of dissolved solid in 125 ml of the solution?
82.8 g
367.2 g
2445.7 g
36.7 g
a solution is found to have an h+ ion concentration 10 000 times lower than that of pure water. which of the following conclusions would correspond to this observation?
a basic solution with a ph of 11
an acidic solution with a ph of 3
an acidic solution with a ph of 11
a basic solution with a ph of 3
First Question
Step1: Define % m/v formula
% m/v = $\frac{\text{mass of solute (g)}}{\text{volume of solution (mL)}} \times 100$
Step2: Rearrange for solute mass
$\text{mass} = \frac{\% \text{ m/v} \times \text{volume}}{100}$
Step3: Substitute given values
$\text{mass} = \frac{29.36 \times 125}{100}$
Step4: Calculate final mass
$\text{mass} = 36.7$
Pure water has $[\text{H}^+] = 1 \times 10^{-7}\ \text{M}$. A concentration 10,000 times lower is $1 \times 10^{-11}\ \text{M}$. pH is calculated as $-\log[\text{H}^+]$, so $\text{pH} = -\log(1 \times 10^{-11}) = 11$. Solutions with pH >7 are basic.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
36.7 g
---