QUESTION IMAGE
Question
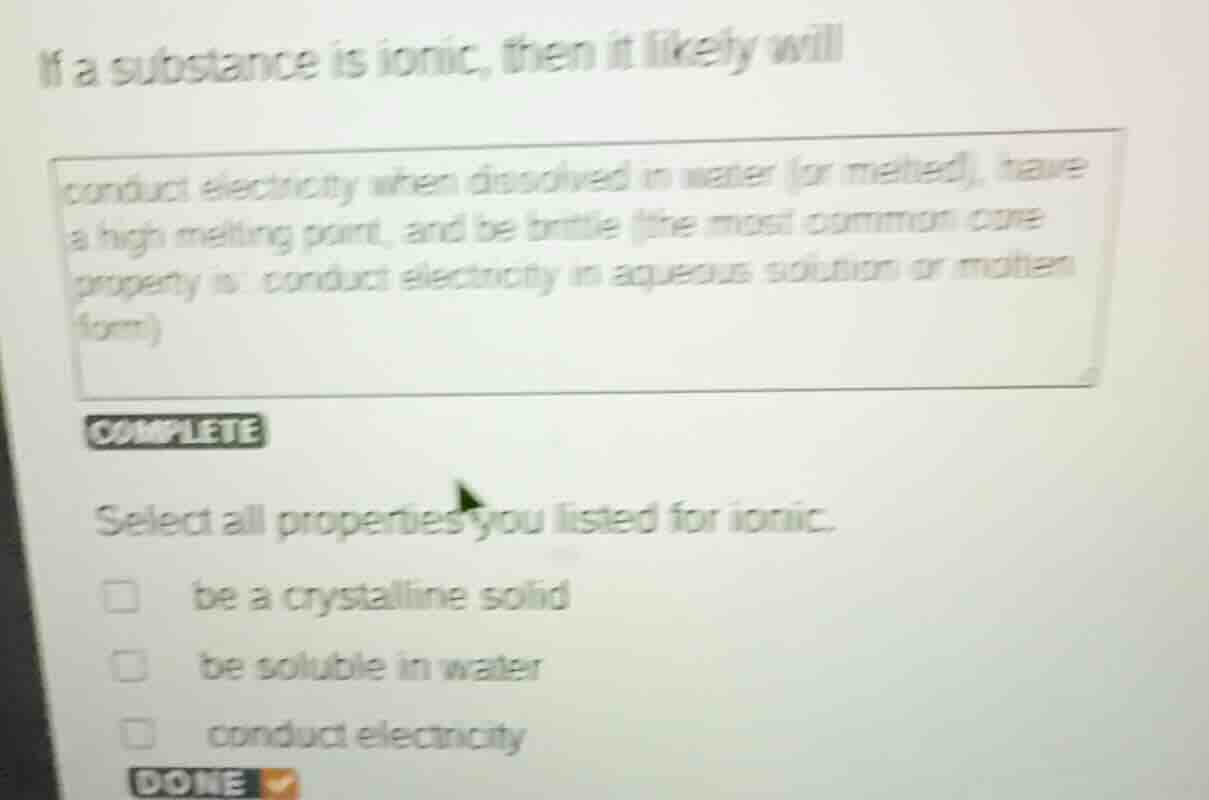
if a substance is ionic, then it likely will conduct electricity when dissolved in water (or melted), have a high melting point, and be brittle (the most common core property is: conduct electricity in aqueous solution or molten form) complete select all properties you listed for ionic. □ be a crystalline solid □ be soluble in water □ conduct electricity done
Ionic compounds form crystalline structures due to their ordered ionic lattice. While some ionic compounds are water-soluble, solubility is not universal for all ionic substances. The key electrical property is conductivity only when dissolved in water or molten, not in solid form.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
☑️ be a crystalline solid
☐ be soluble in water
☐ conduct electricity