QUESTION IMAGE
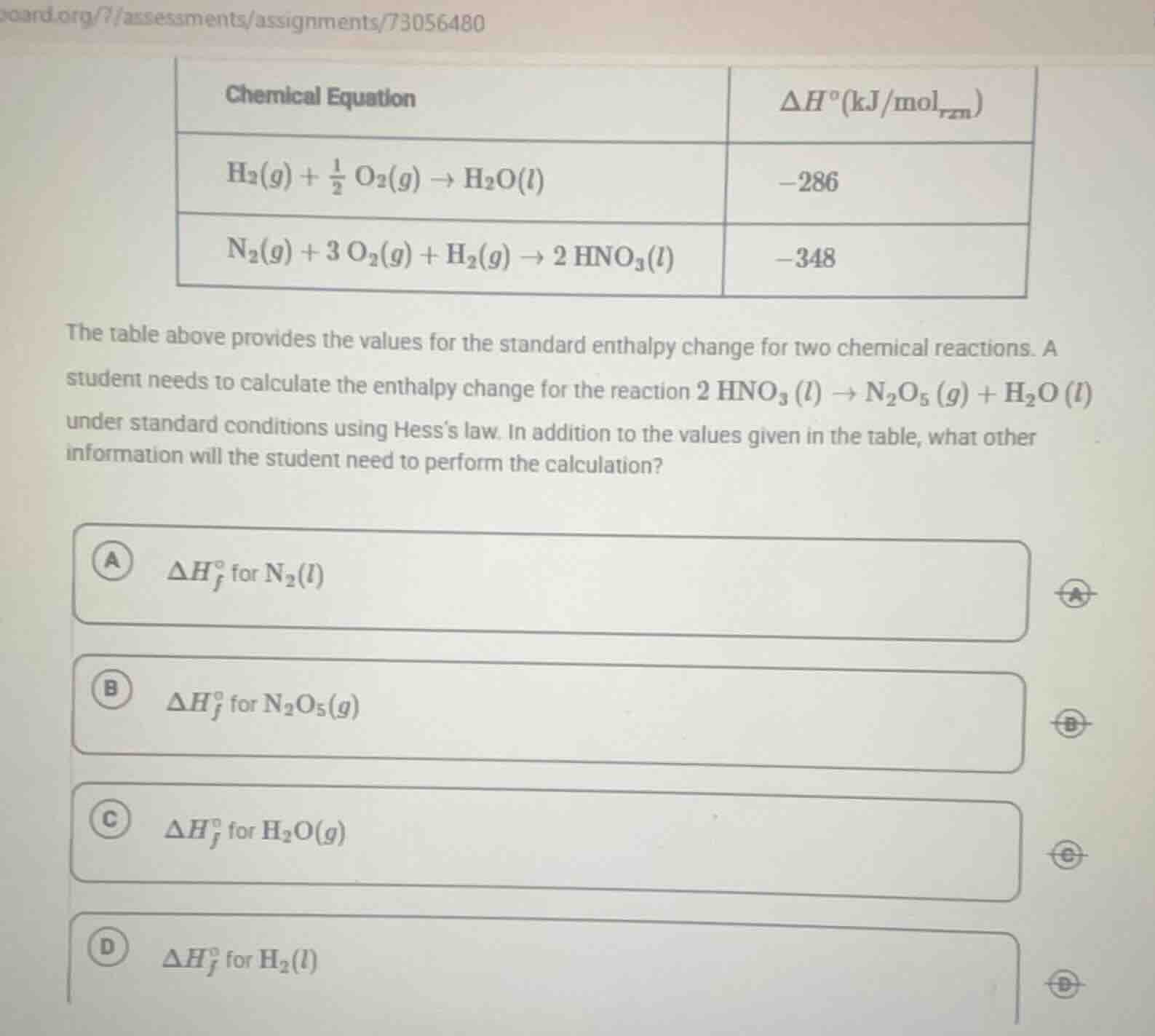
Question
the table above provides the values for the standard enthalpy change for two chemical reactions. a student needs to calculate the enthalpy change for the reaction $2\\ \ce{hno_3}(l)\
ightarrow\ce{n_2o_5}(g)+\ce{h_2o}(l)$ under standard conditions using hesss law. in addition to the values given in the table, what other information will the student need to perform the calculation?\
\
a $\delta h_f^\circ$ for $\ce{n_2}(l)$\
\
b $\delta h_f^\circ$ for $\ce{n_2o_5}(g)$\
\
c $\delta h_f^\circ$ for $\ce{h_2o}(g)$\
\
d $\delta h_f^\circ$ for $\ce{h_2}(l)$
To solve for the enthalpy change of \(2\text{ HNO}_3(l)
ightarrow \text{N}_2\text{O}_5(g) + \text{H}_2\text{O}(l)\) using Hess's law, we analyze the given reactions:
- Reverse the second reaction: \(2\text{ HNO}_3(l)
ightarrow \text{N}_2(g) + 3\text{ O}_2(g) + \text{H}_2(g)\) (enthalpy becomes \(+348\ \text{kJ/mol}\)).
- Keep the first reaction: \(\text{H}_2(g) + \frac{1}{2}\text{ O}_2(g)
ightarrow \text{H}_2\text{O}(l)\) (enthalpy \(-286\ \text{kJ/mol}\)).
- Combine these with the formation of \(\text{N}_2\text{O}_5(g)\) from \(\text{N}_2(g)\) and \(\text{O}_2(g)\): \(\text{N}_2(g) + \frac{5}{2}\text{ O}_2(g)
ightarrow \text{N}_2\text{O}_5(g)\) (requires \(\Delta H_f^\circ\) for \(\text{N}_2\text{O}_5(g)\)).
Options A, C, D involve species (\(\text{N}_2(l)\), \(\text{H}_2\text{O}(g)\), \(\text{H}_2(l)\)) not relevant to the target reaction or given data. Only \(\Delta H_f^\circ\) for \(\text{N}_2\text{O}_5(g)\) (Option B) is needed to complete the Hess’s law calculation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. \(\boldsymbol{\Delta H_f^\circ}\) for \(\boldsymbol{\text{N}_2\text{O}_5(g)}\)