QUESTION IMAGE
Question
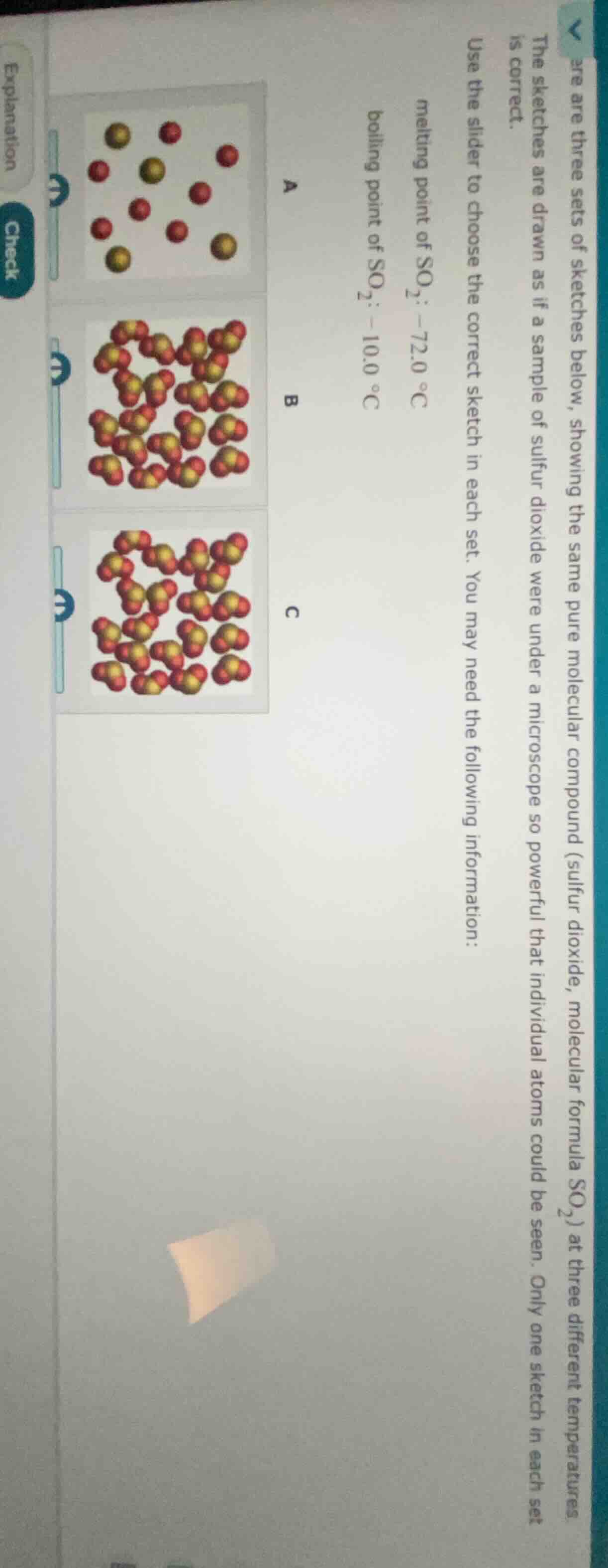
are three sets of sketches below, showing the same pure molecular compound (sulfur dioxide, molecular formula so₂) at three different temperatures. the sketches are drawn as if a sample of sulfur dioxide were under a microscope so powerful that individual atoms could be seen. only one sketch in each set is correct. use the slider to choose the correct sketch in each set. you may need the following information: melting point of so₂: -72.0 °c boiling point of so₂: -10.0 °c
To solve this, we analyze the states of matter based on the melting and boiling points of \( \text{SO}_2 \) (melting point \( -72.0^\circ\text{C} \), boiling point \( -10.0^\circ\text{C} \)):
For Sketch A:
- Atoms are separated (not in molecules), so this represents a gas (particles far apart, moving freely). For \( \text{SO}_2 \), gas state occurs above \( -10.0^\circ\text{C} \) (boiling point).
For Sketch B:
- Molecules are closely packed in an orderly arrangement, typical of a solid (particles vibrate in fixed positions). For \( \text{SO}_2 \), solid state occurs below \( -72.0^\circ\text{C} \) (melting point).
For Sketch C:
- Molecules are close but less orderly than a solid, typical of a liquid (particles slide past each other). For \( \text{SO}_2 \), liquid state occurs between \( -72.0^\circ\text{C} \) (melting point) and \( -10.0^\circ\text{C} \) (boiling point).
Final Answers (assuming the question asks to identify each sketch’s state context):
- Sketch A: Gas (matches \( T > -10.0^\circ\text{C} \))
- Sketch B: Solid (matches \( T < -72.0^\circ\text{C} \))
- Sketch C: Liquid (matches \( -72.0^\circ\text{C} < T < -10.0^\circ\text{C} \))
(If the question specifies choosing one per “set” or context, clarify the temperature range, but the above explains the states for each sketch.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze the states of matter based on the melting and boiling points of \( \text{SO}_2 \) (melting point \( -72.0^\circ\text{C} \), boiling point \( -10.0^\circ\text{C} \)):
For Sketch A:
- Atoms are separated (not in molecules), so this represents a gas (particles far apart, moving freely). For \( \text{SO}_2 \), gas state occurs above \( -10.0^\circ\text{C} \) (boiling point).
For Sketch B:
- Molecules are closely packed in an orderly arrangement, typical of a solid (particles vibrate in fixed positions). For \( \text{SO}_2 \), solid state occurs below \( -72.0^\circ\text{C} \) (melting point).
For Sketch C:
- Molecules are close but less orderly than a solid, typical of a liquid (particles slide past each other). For \( \text{SO}_2 \), liquid state occurs between \( -72.0^\circ\text{C} \) (melting point) and \( -10.0^\circ\text{C} \) (boiling point).
Final Answers (assuming the question asks to identify each sketch’s state context):
- Sketch A: Gas (matches \( T > -10.0^\circ\text{C} \))
- Sketch B: Solid (matches \( T < -72.0^\circ\text{C} \))
- Sketch C: Liquid (matches \( -72.0^\circ\text{C} < T < -10.0^\circ\text{C} \))
(If the question specifies choosing one per “set” or context, clarify the temperature range, but the above explains the states for each sketch.)