QUESTION IMAGE
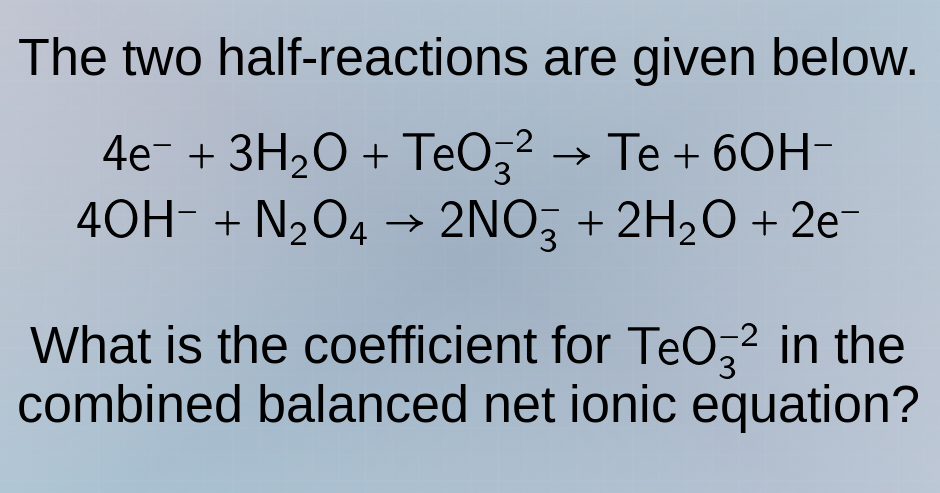
Question
the two half - reactions are given below.
$4e^- + 3h_2o + teo_3^{-2} \to te + 6oh^-$
$4oh^- + n_2o_4 \to 2no_3^- + 2h_2o + 2e^-$
what is the coefficient for $teo_3^{-2}$ in the combined balanced net ionic equation?
Step1: Balance electrons in half-reactions
The first half-reaction has \(4e^-\) gained, and the second has \(2e^-\) lost. Multiply the second half-reaction by 2 to balance electrons:
\(2\times(4OH^- + N_2O_4
ightarrow 2NO_3^- + 2H_2O + 2e^-)\)
Gives: \(8OH^- + 2N_2O_4
ightarrow 4NO_3^- + 4H_2O + 4e^-\)
Step2: Add the two half-reactions
First half-reaction: \(4e^- + 3H_2O + TeO_3^{2-}
ightarrow Te + 6OH^-\)
Second (multiplied) half-reaction: \(8OH^- + 2N_2O_4
ightarrow 4NO_3^- + 4H_2O + 4e^-\)
Add them together, cancel \(4e^-\) and simplify \(H_2O\) and \(OH^-\):
\(4e^- + 3H_2O + TeO_3^{2-} + 8OH^- + 2N_2O_4
ightarrow Te + 6OH^- + 4NO_3^- + 4H_2O + 4e^-\)
Simplify \(H_2O\) (4 - 3 = 1 left on product side) and \(OH^-\) (8 - 6 = 2 left on reactant side):
\(TeO_3^{2-} + 2OH^- + 2N_2O_4
ightarrow Te + 4NO_3^- + H_2O\)
Step3: Identify the coefficient of \(TeO_3^{2-}\)
From the balanced equation, the coefficient of \(TeO_3^{2-}\) is 1. Wait, no—wait, did I make a mistake? Wait, no, let's recheck. Wait, original first half-reaction: \(TeO_3^{2-}\) has coefficient 1, and after balancing electrons (multiplying second by 2), when adding, the \(TeO_3^{2-}\) remains with coefficient 1? Wait, no, wait: Wait, the first half-reaction is \(4e^- + 3H_2O + TeO_3^{2-}
ightarrow Te + 6OH^-\) (coefficient 1 for \(TeO_3^{2-}\)). The second half-reaction is multiplied by 2, but the first is not multiplied. So when adding, the \(TeO_3^{2-}\) has coefficient 1? Wait, no, wait, let's redo the addition:
First half-reaction (reactants: \(4e^-, 3H_2O, TeO_3^{2-}\); products: \(Te, 6OH^-\))
Second half-reaction (after multiplying by 2: reactants: \(8OH^-, 2N_2O_4\); products: \(4NO_3^-, 4H_2O, 4e^-\))
Adding:
Reactants: \(4e^-, 3H_2O, TeO_3^{2-}, 8OH^-, 2N_2O_4\)
Products: \(Te, 6OH^-, 4NO_3^-, 4H_2O, 4e^-\)
Cancel \(4e^-\):
Reactants: \(3H_2O, TeO_3^{2-}, 8OH^-, 2N_2O_4\)
Products: \(Te, 6OH^-, 4NO_3^-, 4H_2O\)
Now, \(H_2O\): 4 (product) - 3 (reactant) = 1 (product)
\(OH^-\): 8 (reactant) - 6 (product) = 2 (reactant)
So balanced equation: \(TeO_3^{2-} + 2OH^- + 2N_2O_4
ightarrow Te + 4NO_3^- + H_2O\)
Wait, but the coefficient of \(TeO_3^{2-}\) is 1? But that seems off. Wait, no, wait, maybe I messed up the electron balance. Wait, first half-reaction: Te goes from +4 (in \(TeO_3^{2-}\): Te + 3*(-2) = -2 → Te = +4) to 0 (in Te), so gain 4e^- (correct). Second half-reaction: N in \(N_2O_4\): each N is +4, in \(NO_3^-\) N is +5, so each N loses 1e^-, two Ns lose 2e^- per \(N_2O_4\), so per reaction, 2e^- lost (correct). So multiplying second by 2 to get 4e^- lost, matches 4e^- gained. Then adding:
First half-reaction: \(TeO_3^{2-} + 3H_2O + 4e^-
ightarrow Te + 6OH^-\)
Second (x2): \(2N_2O_4 + 8OH^-
ightarrow 4NO_3^- + 4H_2O + 4e^-\)
Add: \(TeO_3^{2-} + 3H_2O + 4e^- + 2N_2O_4 + 8OH^-
ightarrow Te + 6OH^- + 4NO_3^- + 4H_2O + 4e^-\)
Cancel \(4e^-\), \(H_2O\) (4-3=1), \(OH^-\) (8-6=2):
\(TeO_3^{2-} + 2OH^- + 2N_2O_4
ightarrow Te + 4NO_3^- + H_2O\)
Yes, so the coefficient of \(TeO_3^{2-}\) is 1? Wait, but that seems correct? Wait, no, wait, maybe I made a mistake in the first half-reaction's coefficient. Wait, the first half-reaction is given as \(4e^- + 3H_2O + TeO_3^{2-}
ightarrow Te + 6OH^-\), so the coefficient of \(TeO_3^{2-}\) is 1 in the original, and when combining, since we didn't multiply the first half-reaction, its coefficient remains 1. Wait, but let's check the electron balance again. The first half-reaction gains 4e^-, the second (original) loses 2e^-, so to balance, multiply the second by 2 (loses 4e^-), so when adding, the first (1…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1