QUESTION IMAGE
Question
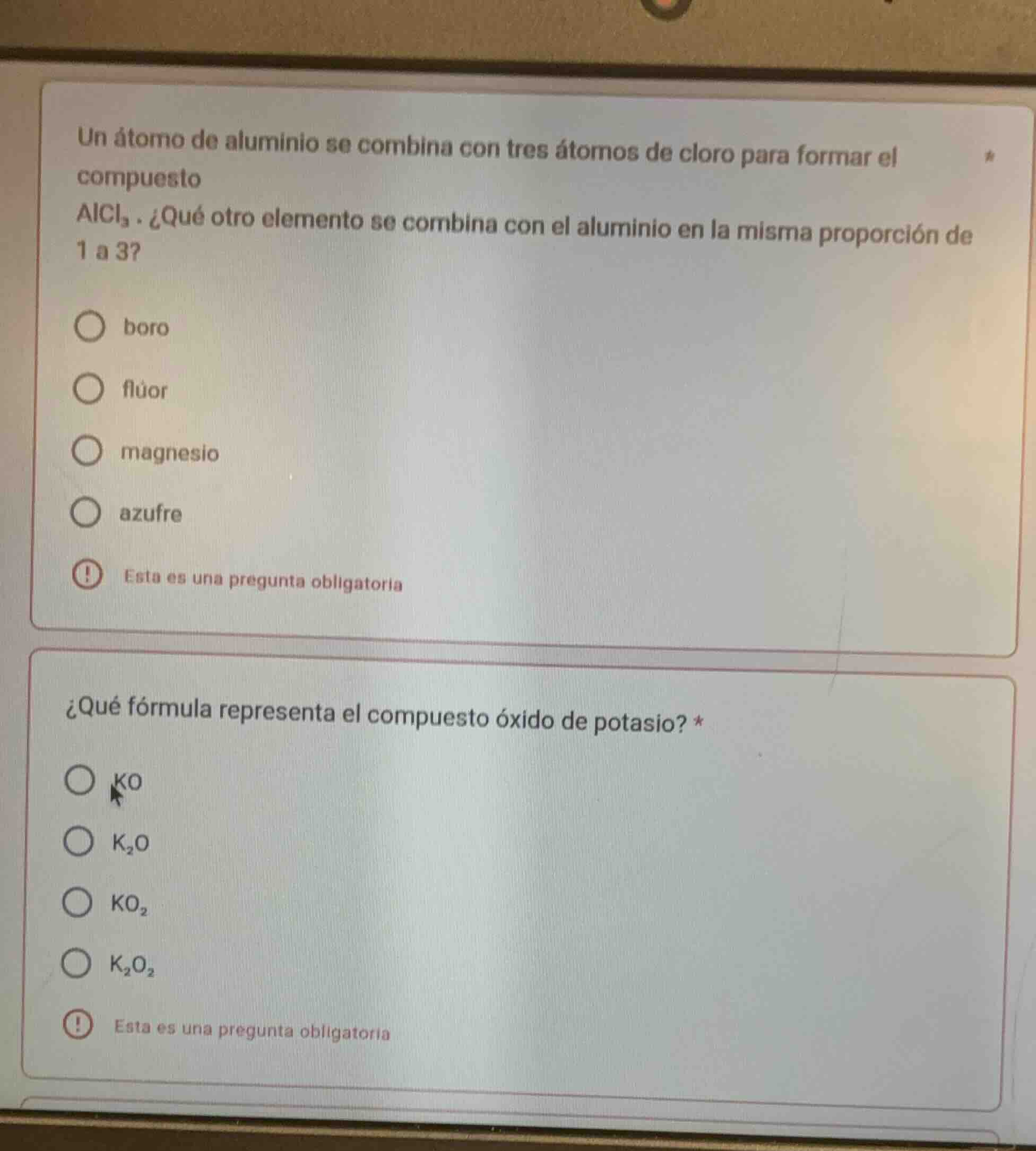
un átomo de aluminio se combina con tres átomos de cloro para formar el compuesto
alcl₃. ¿qué otro elemento se combina con el aluminio en la misma proporción de
1 a 3?
○ boro
○ flúor
○ magnesio
○ azufre
! esta es una pregunta obligatoria
¿qué fórmula representa el compuesto óxido de potasio? *
○ ko
○ k₂o
○ ko₂
○ k₂o₂
! esta es una pregunta obligatoria
First Question (Element combining with Aluminum in 1:3 ratio)
To determine which element combines with aluminum in a 1:3 ratio, we analyze the valence of aluminum and the elements. Aluminum has a valence of +3 (since in \( \text{AlCl}_3 \), Cl has -1, so 3 Cl⁻ balance Al³⁺). Fluorine (flúor) also has a valence of -1 (like Cl), so aluminum (Al³⁺) will combine with fluorine (F⁻) in a 1:3 ratio to form \( \text{AlF}_3 \). Boron is a non - metal with different bonding, magnesium is +2, and sulfur is -2 (so ratio would be different).
Potassium (K) has a valence of +1, and oxygen (O) in oxide has a valence of -2. To balance the charges, we use the criss - cross method. The number of K⁺ ions needed is 2 (since \( 2\times( + 1)=+2 \)) and the number of O²⁻ ions needed is 1 (since \( 1\times(-2)= - 2 \)). So the formula is \( \text{K}_2\text{O} \). \( \text{KO} \) has unbalanced charges, \( \text{KO}_2 \) is potassium superoxide, and \( \text{K}_2\text{O}_2 \) is potassium peroxide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. flúor