QUESTION IMAGE
Question
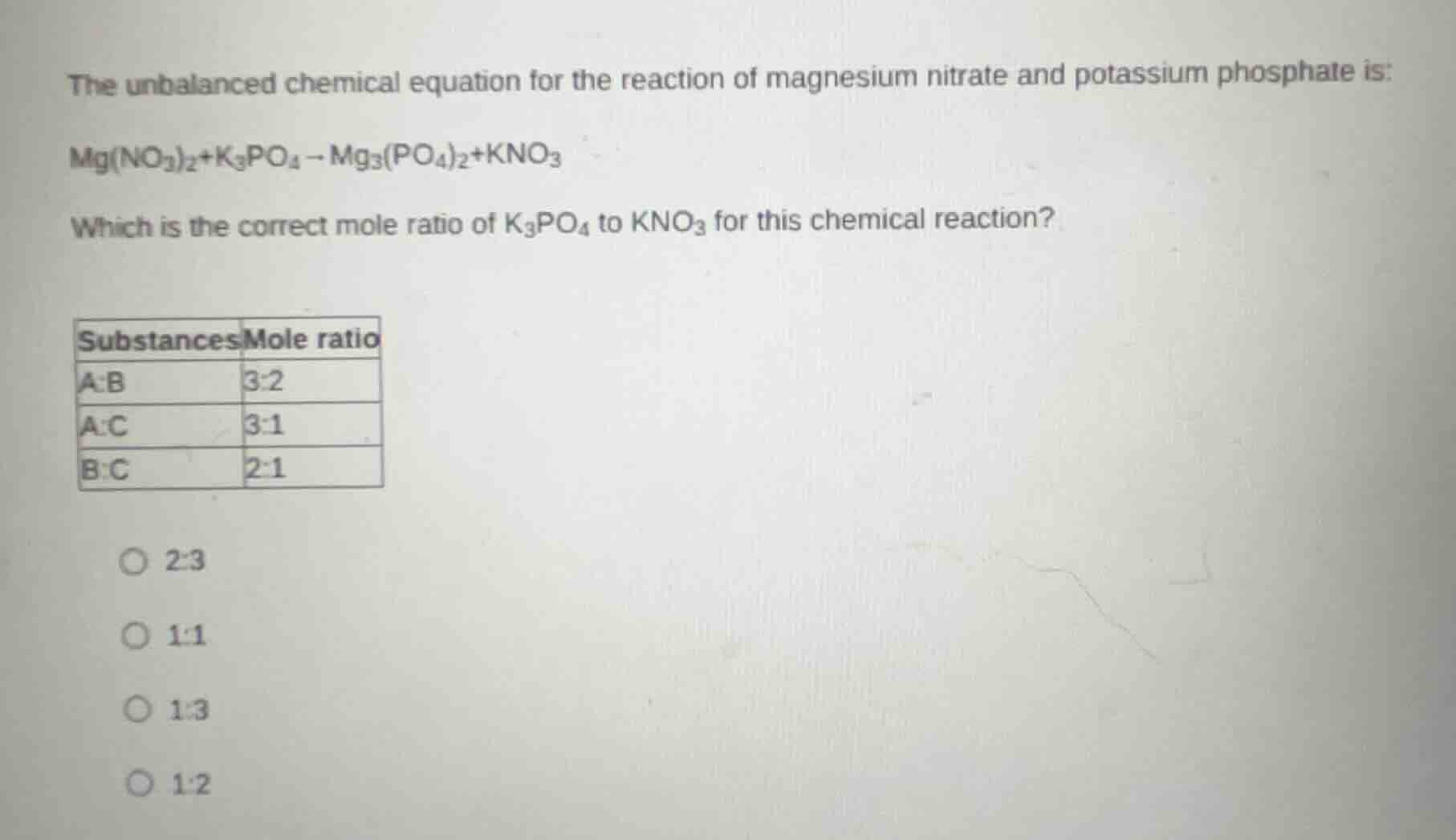
the unbalanced chemical equation for the reaction of magnesium nitrate and potassium phosphate is:
mg(no₃)₂+k₃po₄→mg₃(po₄)₂+kno₃
which is the correct mole ratio of k₃po₄ to kno₃ for this chemical reaction?
substances mole ratio
a:b 3:2
a:c 3:1
b:c 2:1
○ 2:3
○ 1:1
○ 1:3
○ 1:2
Step1: Balance the chemical equation
First, balance the magnesium (Mg) atoms. On the left, we have 1 Mg in $Mg(NO_3)_2$, and on the right, we have 3 Mg in $Mg_3(PO_4)_2$. So we put a coefficient of 3 in front of $Mg(NO_3)_2$:
Next, balance the phosphate ($PO_4^{3 - }$) groups. On the left, we have 1 $PO_4^{3 - }$ in $K_3PO_4$, and on the right, we have 2 $PO_4^{3 - }$ in $Mg_3(PO_4)_2$. So we put a coefficient of 2 in front of $K_3PO_4$:
Now, balance the potassium (K) atoms. On the left, we have 2 * 3 = 6 K in $2K_3PO_4$, so we need 6 K on the right. We put a coefficient of 6 in front of $KNO_3$:
Let's check the nitrate ($NO_3^ - $) groups. On the left, we have 3 * 2 = 6 $NO_3^ - $ in $3Mg(NO_3)_2$, and on the right, we have 6 $NO_3^ - $ in $6KNO_3$. So the equation is balanced:
$$3Mg(NO_3)_2 + 2K_3PO_4 = Mg_3(PO_4)_2 + 6KNO_3$$
Step2: Determine the mole ratio of $K_3PO_4$ to $KNO_3$
From the balanced equation, the coefficient of $K_3PO_4$ is 2, and the coefficient of $KNO_3$ is 6. So the mole ratio of $K_3PO_4$ to $KNO_3$ is 2:6, which simplifies to 1:3.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1:3