QUESTION IMAGE
Question
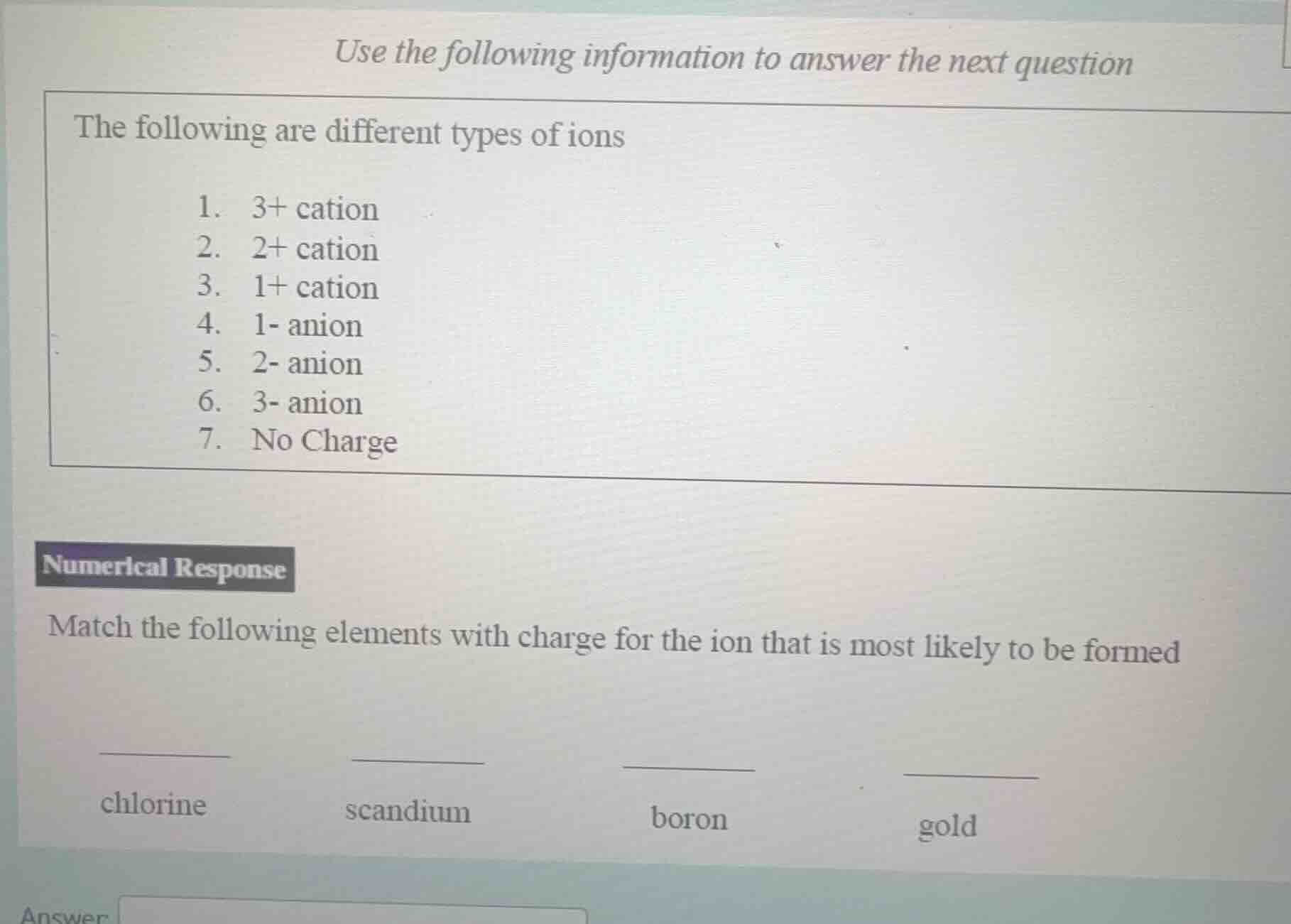
use the following information to answer the next question
the following are different types of ions
- 3+ cation
- 2+ cation
- 1+ cation
- 1- anion
- 2- anion
- 3- anion
- no charge
numerical response
match the following elements with charge for the ion that is most likely to be formed
chlorine scandium boron gold
Step1: Match chlorine to ion type
Chlorine (Group 17) gains 1 electron → 1- anion (4)
Step2: Match scandium to ion type
Scandium (Group 3) loses 3 electrons → 3+ cation (1)
Step3: Match boron to ion type
Boron (Group 13) loses 3 electrons → 3+ cation (1)
Step4: Match gold to ion type
Gold commonly forms 1+ cation (3)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
chlorine: 4, scandium: 1, boron: 1, gold: 3