QUESTION IMAGE
Question
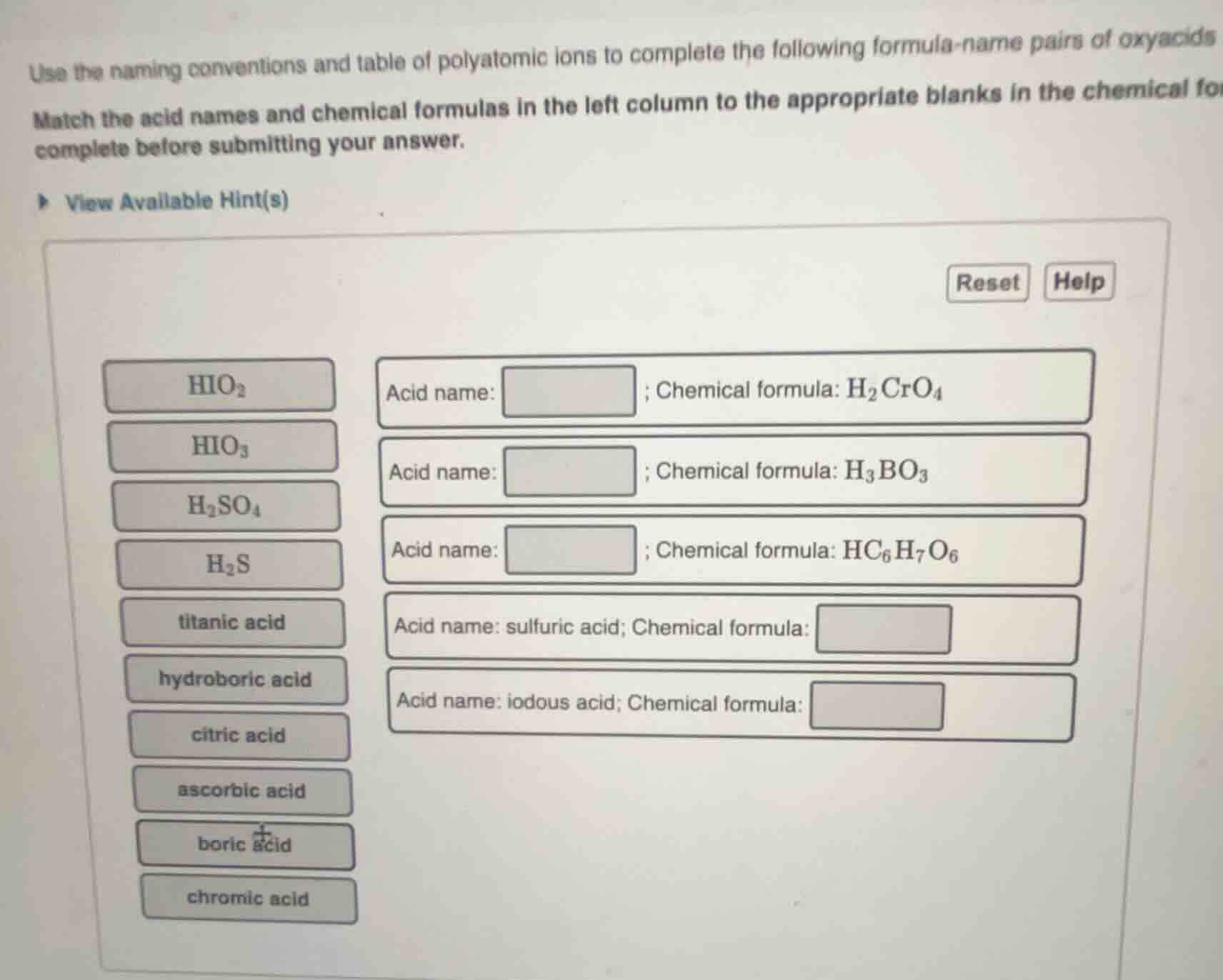
use the naming conventions and table of polyatomic ions to complete the following formula-name pairs of oxyacids
match the acid names and chemical formulas in the left column to the appropriate blanks in the chemical formula complete before submitting your answer.
view available hint(s)
reset help
$ce{hio_{2}}$
$ce{hio_{3}}$
$ce{h_{2}so_{4}}$
$ce{h_{2}s}$
titanic acid
hydroboric acid
citric acid
ascorbic acid
boric acid
chromic acid
acid name: ; chemical formula: $ce{h_{2}cro_{4}}$
acid name: ; chemical formula: $ce{h_{3}bo_{3}}$
acid name: ; chemical formula: $ce{hc_{6}h_{7}o_{6}}$
acid name: sulfuric acid; chemical formula:
acid name: iodous acid; chemical formula:
- For $\ce{H2CrO4}$, the polyatomic ion is chromate ($\ce{CrO4^2-}$), so the acid is named chromic acid.
- For $\ce{H3BO3}$, the polyatomic ion is borate ($\ce{BO3^3-}$), so the acid is named boric acid.
- For $\ce{HC6H7O6}$, this is the formula for ascorbic acid (vitamin C).
- Sulfuric acid corresponds to the formula $\ce{H2SO4}$, as it contains the sulfate ion ($\ce{SO4^2-}$) with two hydrogen ions to balance charge.
- Iodous acid comes from the iodite ion ($\ce{IO2^-}$), so its formula is $\ce{HIO2}$ (the "-ous" suffix indicates the lower oxidation state iodine polyatomic ion).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Acid name: chromic acid; Chemical formula: $\ce{H2CrO4}$
- Acid name: boric acid; Chemical formula: $\ce{H3BO3}$
- Acid name: ascorbic acid; Chemical formula: $\ce{HC6H7O6}$
- Acid name: sulfuric acid; Chemical formula: $\ce{H2SO4}$
- Acid name: iodous acid; Chemical formula: $\ce{HIO2}$