QUESTION IMAGE
Question
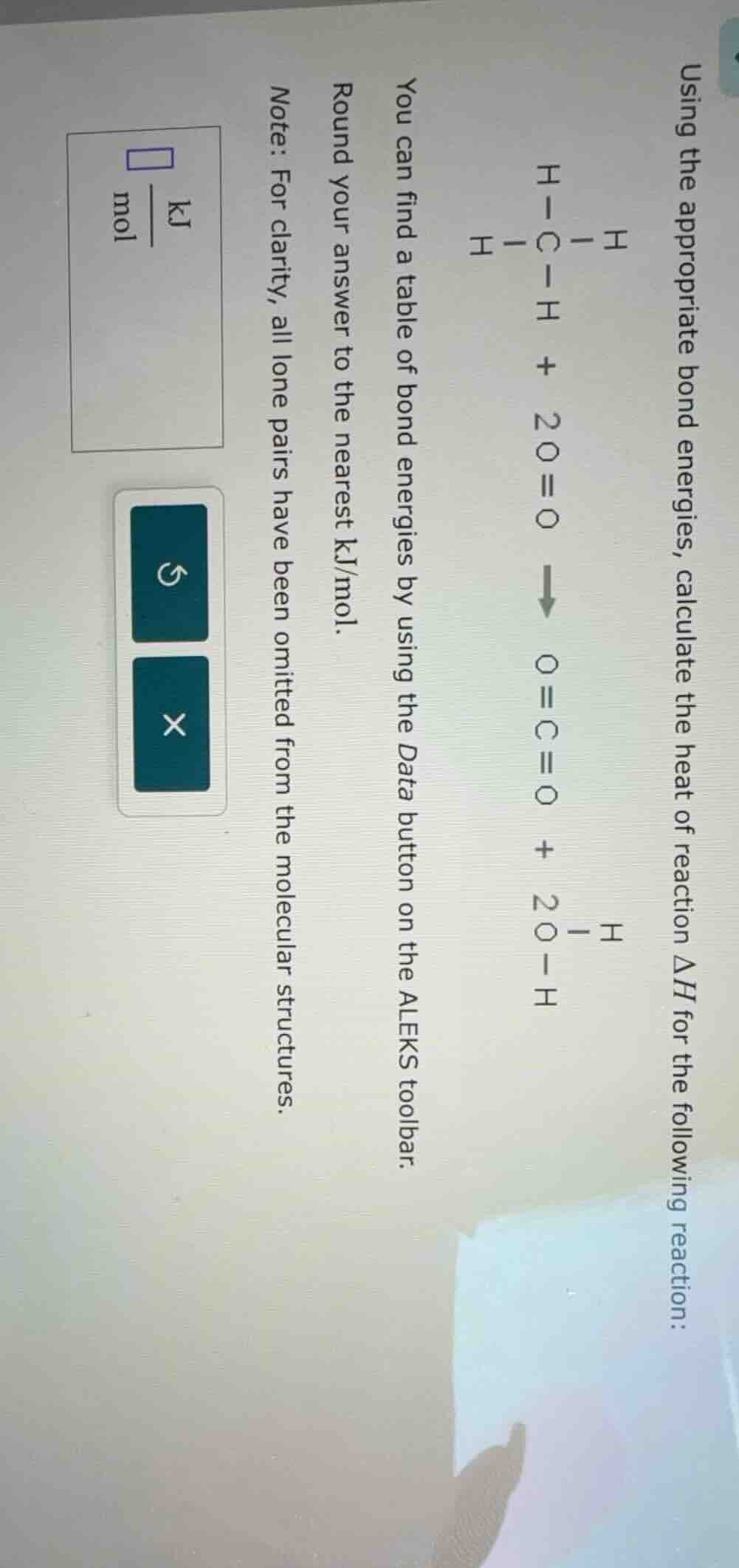
using the appropriate bond energies, calculate the heat of reaction \\(\delta h\\) for the following reaction:
\\(\ce{h - c - h}) (with h on the left, right, and below c) \\(\ce{ + 2 o=o -> o=c=o + 2 o - h}) (with h on the left of o)
you can find a table of bond energies by using the data button on the aleks toolbar.
round your answer to the nearest kj/mol.
note: for clarity, all lone pairs have been omitted from the molecular structures.
(box with \\(\frac{\text{kj}}{\text{mol}}\\) to fill in, and two buttons: a refresh and an x)
Step1: Identify bonds in reactants
Reactants: 1 molecule of $\text{CH}_4$ (4 $\text{C-H}$ bonds) + 2 molecules of $\text{O}_2$ (2 $\text{O=O}$ bonds)
Bond energies (standard values): $\text{C-H} = 413\ \text{kJ/mol}$, $\text{O=O} = 498\ \text{kJ/mol}$
Step2: Calculate energy to break bonds
Energy in = $(4 \times 413) + (2 \times 498)$
$= 1652 + 996 = 2648\ \text{kJ/mol}$
Step3: Identify bonds in products
Products: 1 molecule of $\text{CO}_2$ (2 $\text{C=O}$ bonds) + 2 molecules of $\text{H}_2\text{O}$ (4 $\text{O-H}$ bonds)
Bond energies (standard values): $\text{C=O} = 799\ \text{kJ/mol}$, $\text{O-H} = 464\ \text{kJ/mol}$
Step4: Calculate energy from forming bonds
Energy out = $(2 \times 799) + (4 \times 464)$
$= 1598 + 1856 = 3454\ \text{kJ/mol}$
Step5: Compute reaction enthalpy
$\Delta H = \text{Energy in} - \text{Energy out}$
$\Delta H = 2648 - 3454$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$-806\ \frac{\text{kJ}}{\text{mol}}$