QUESTION IMAGE
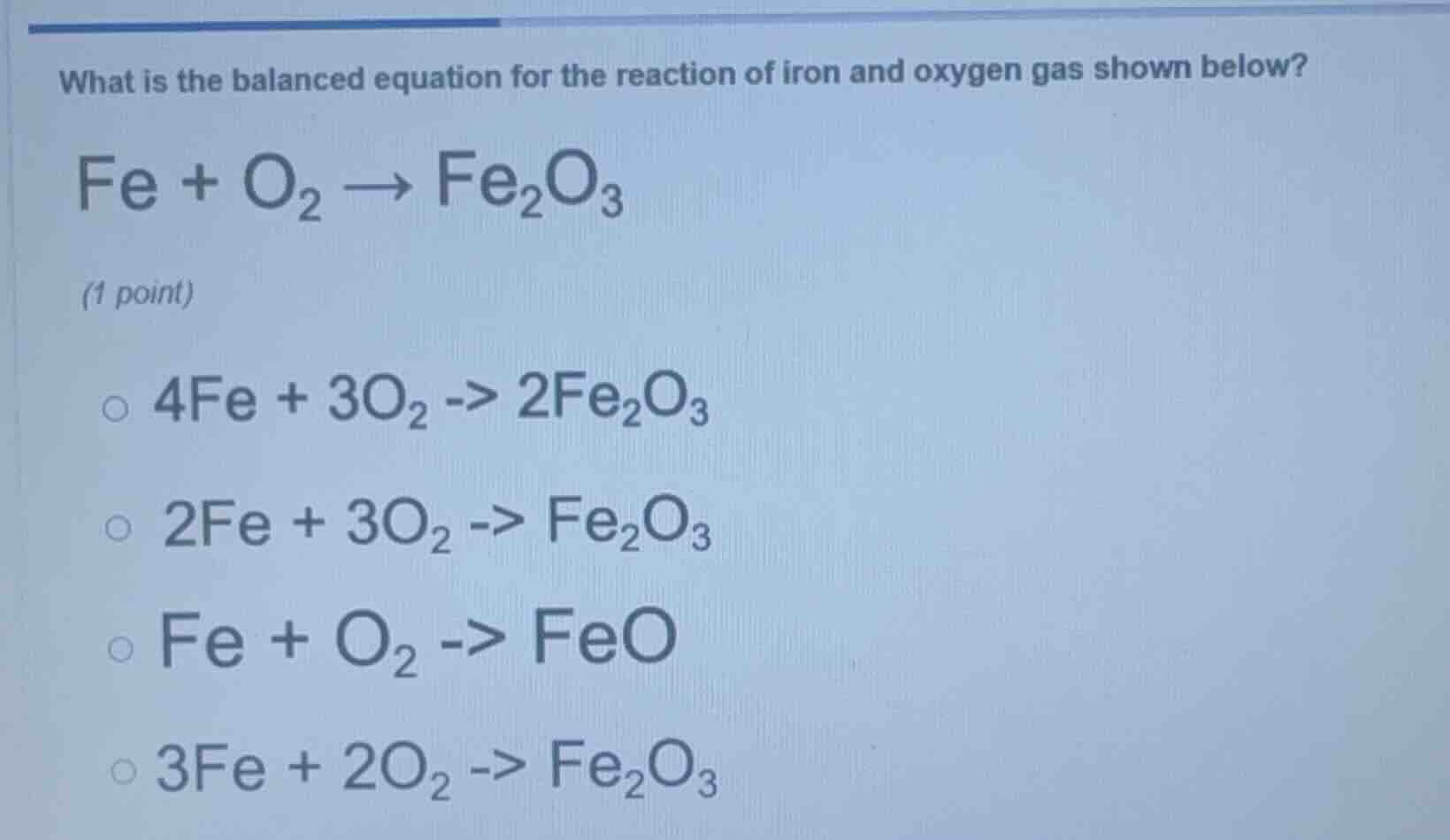
Question
what is the balanced equation for the reaction of iron and oxygen gas shown below? fe + o₂ → fe₂o₃ (1 point) 4fe + 3o₂ -> 2fe₂o₃ 2fe + 3o₂ -> fe₂o₃ fe + o₂ -> feo 3fe + 2o₂ -> fe₂o₃
Step1: Check Fe atoms
On left: 4 Fe. On right: \(2\times2 = 4\) Fe. Balanced.
Step2: Check O atoms
On left: \(3\times2 = 6\) O. On right: \(2\times3 = 6\) O. Balanced.
Other options:
- \(2\text{Fe} + 3\text{O}_2 \to \text{Fe}_2\text{O}_3\): O on left \(6\), right \(3\) – unbalanced.
- \(\text{Fe} + \text{O}_2 \to \text{FeO}\): Product wrong, O unbalanced.
- \(3\text{Fe} + 2\text{O}_2 \to \text{Fe}_2\text{O}_3\): Fe on left \(3\), right \(2\) – unbalanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \(4\text{Fe} + 3\text{O}_2 \to 2\text{Fe}_2\text{O}_3\)