QUESTION IMAGE
Question
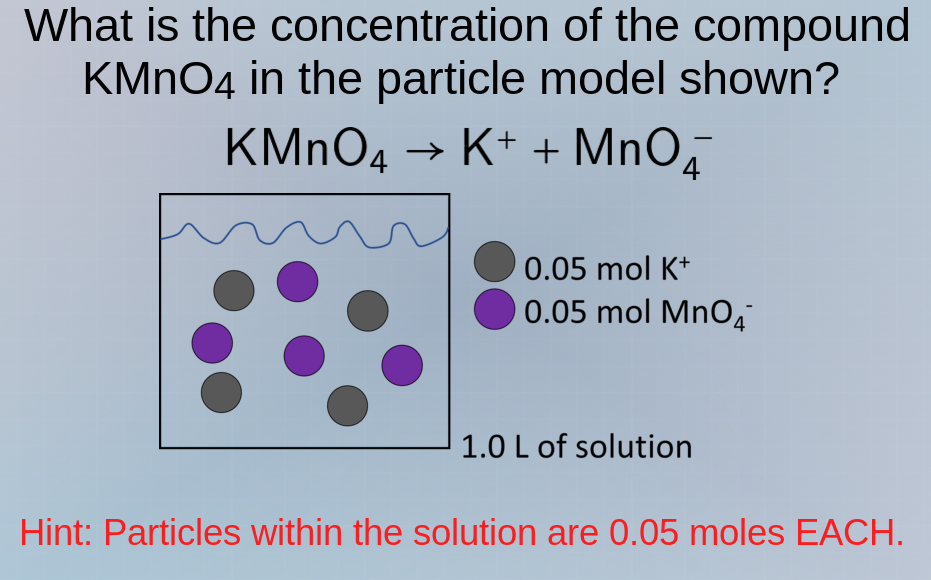
what is the concentration of the compound kmno₄ in the particle model shown? kmno₄ → k⁺ + mno₄⁻ hint: particles within the solution are 0.05 moles each. 0.05 mol k⁺ 0.05 mol mno₄⁻ 1.0 l of solution
Step1: Count moles of \( K^+ \)
From the diagram, there are 4 gray circles ( \( K^+ \) ). Each is 0.05 mol.
Moles of \( K^+ = 4 \times 0.05\ \text{mol} = 0.2\ \text{mol} \).
Step2: Count moles of \( \text{MnO}_4^- \)
There are 4 purple circles ( \( \text{MnO}_4^- \) ). Each is 0.05 mol.
Moles of \( \text{MnO}_4^- = 4 \times 0.05\ \text{mol} = 0.2\ \text{mol} \).
Step3: Determine moles of \( \text{KMnO}_4 \)
From the dissociation \( \text{KMnO}_4
ightarrow \text{K}^+ + \text{MnO}_4^- \), 1 mol \( \text{KMnO}_4 \) produces 1 mol \( \text{K}^+ \) and 1 mol \( \text{MnO}_4^- \).
Moles of \( \text{KMnO}_4 \) = moles of \( \text{K}^+ \) (or \( \text{MnO}_4^- \)) = 0.2 mol.
Step4: Calculate concentration
Concentration \( = \frac{\text{moles of solute}}{\text{volume of solution (L)}} \).
Volume = 1.0 L.
Concentration of \( \text{KMnO}_4 = \frac{0.2\ \text{mol}}{1.0\ \text{L}} = 0.2\ \text{M} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The concentration of \( \text{KMnO}_4 \) is \( \boldsymbol{0.2\ \text{M}} \) (or \( 0.2\ \text{mol/L} \)).